The discovery of a brain circuit ‘shortcut’ could explain why some addicts unintentionally relapse, and suggests that a shift in focus for therapies might help those who want to stay off drugs.
There is a road down which those with substance addiction travel. Its beginnings are influenced by circumstances and genetics; it becomes well trodden, habitual, initially reinforced by pleasurable effects and then by cues; and, for some, it will become a road they can never leave.
Addiction is a chronic, relapsing psychiatric disease, with complex behavioural processes and equally complex changes to brain circuits. The brain of a person who has drunk alcohol or taken drugs is different to the brain of one who has not, as pharmacologist Dr David Belin describes: “I like whisky. I started drinking whisky when I was 20 and I have only a small amount now and then. But if I were to scan my brain, it changed that very first time, and it continues to be changed.”
And because drugs change the brain in ways that foster compulsive drug and alcohol abuse, quitting is difficult, even for those who want to. Belin and Professor Barry Everitt, from the Department of Pharmacology, have shown that some are more vulnerable than others to developing addiction – and to relapsing.
Their recent research, published in Nature Communications, uncovers a new neural ‘short cut’, or ‘back door’, in rat brains that could explain why some cocaine addicts relapse without intending to. The results, they believe, could suggest new forms of behavioural and pharmacological therapies.
A decade ago, both Belin and Everitt, independently, were the first to show that addiction manifests itself differently in different individuals and that, for some, compulsive cocaine-seeking behaviour would continue despite adverse circumstances. In a rat model, around 20% of animals addicted to cocaine still sought the drug despite the risk of receiving mild electric shocks on doing so.
“This was the first time in the field of addiction that the idea of inter-individual differences in vulnerability to compulsive drug seeking was raised,” explains Belin, who is funded by the Wellcome Trust. “With regard to psychiatric disorders, we are not equally vulnerable.”
Drug addiction had largely been regarded as the end point of a progressive loss of control over drug seeking resulting from a failure of part of the brain – the prefrontal cortex – that deals with decision making.
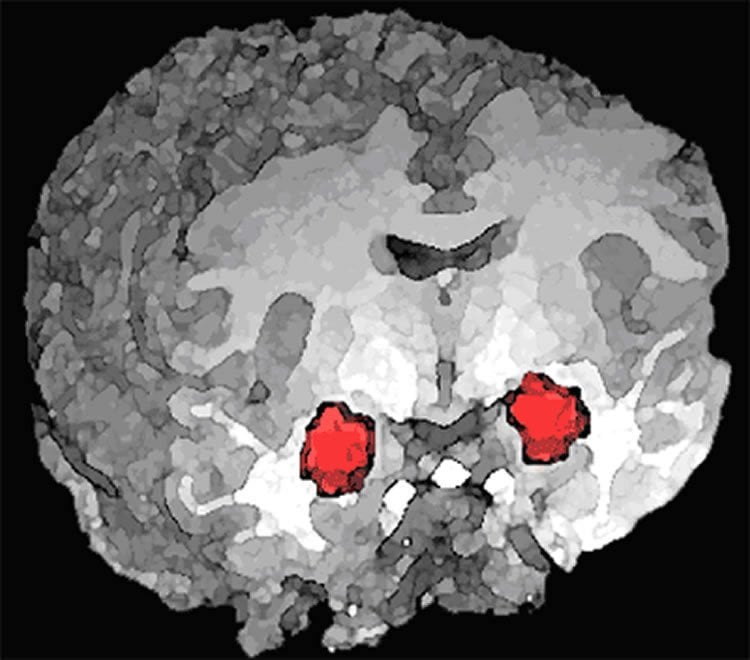
“Certainly, chronic exposure to drugs alters the prefrontal cortex, which governs motivation, inhibitory control and choice. But it also alters an area of the brain called the basolateral amygdala, which is associated with the link between a stimulus and an emotion,” explains Belin.
To explain further he gives the example of choosing to eat a cake or an apple. “The basolateral amygdala stores the pleasurable memories associated with eating the cake, but the prefrontal cortex manipulates this information, helping you to weigh up whether or not you should eat another slice or choose the apple instead. If you eat the slice, regions of the ventral striatum, the structure that processes reward and links emotions to actions, are activated.”
However, Belin and Everitt’s latest research has shown that this is not the only neuronal circuit that underlies cocaine-seeking behaviour.
Using their rat model, they identified a completely new highway that links impulses with habits. This brain circuit links the basolateral amygdala indirectly with the dorsolateral striatum, which is the neural locus of habits.
“Because it doesn’t recruit the prefrontal cortex it doesn’t involve choice,” explains Belin. “It’s a short cut or back door directly to habit. It means that addicts can have internal urges they are not aware of that drive drug seeking. It’s a newly recognised function of this brain circuitry.
“It would explain situations in which individuals who have been abstinent for five years suddenly relapse, telling their counsellor ‘I was walking in the street and I found myself with a glass of wine – and I promise you I didn’t want it.’ This has often been dismissed as ‘weakness of the will’ and then denial. This may also happen, but what our results in rats suggest is that there are occasions in which the stimuli go via motivation straight to the habit part of the brain without an individual even being aware of it.”
The researchers believe that this is a breakthrough in understanding how drugs like cocaine can hijack the brain with such devastating consequences. “We can speculate that the subset of individuals currently using drugs who are especially vulnerable to addiction might have a stronger pathway – a ‘superhighway’ to habit formation.”
The finding could explain the puzzling feature of why substance abusers repeatedly do something they know is bad for them. This is an area that interests Dr Valerie Voon in the Department of Psychiatry. “There appears to be a form of impulsivity called ‘waiting impulsivity’ that doesn’t involve choice and that predisposes to drug addiction.”
Waiting impulsivity is seen when a runner takes off before the starter pistol, or when someone interrupts inappropriately. Voon has devised a way of measuring it in humans based on tasks studied in rats. She also uses another task that teases apart decisions that are made through choices that are goal directed (i.e. taking into account the goal or outcome) from those that are habitual (i.e. relying rather automatically on past rewarding choices).
“We find that alcohol addiction correlates with waiting impulsivity. Once you’re abstinent, the compulsivity or habits also improve. Now that we have this test we can start to cut across other addictions – is there an underlying neural process shared by all? Can we ascribe causality? Can we suggest new treatments?”
Belin suggests that a combination of behavioural and pharmacological treatments might be the answer to helping addicts quit.
Treatments such as cognitive behavioural therapy aim to restore the function of the prefrontal cortex so that emotions don’t automatically elicit habits. “But if people are not aware of their impulses then they can’t subjectively or cognitively apprehend the motivation and the impulse to take drugs or drink,” says Belin. “It’s possible that mindfulness might be beneficial in helping them identify the impulses.”
Although there is currently no effective pharmacological treatment for cocaine addiction, his team has recently shown that N-acetylcysteine might be a possibility, but only if used early enough on the road to addiction when the individual still has the motivation to stop.
The researchers now plan to extend their study of cocaine addiction to compulsive alcohol- and heroin-seeking behaviour. They and Voon would like to understand to what degree behavioural traits such as impulsivity, novelty preference and anxiety – using brain imaging to identify neural correlates – can be used as a predictor of compulsive drug seeking.
“Drug taking is always volitional to begin with – you take drugs because you want to experience something,” adds Belin. “Unfortunately they hijack the learning mechanisms in your brain so that you don’t really take them because you want them but because stimuli in your environment tell you to do so. We want to work out how we can help people become aware that their impulses are wrongly triggering their habits.”
Source: Craig Brierley – University of Cambridge
Image Source: The image is in the public domain.
Original Research: Full open access research for “Basolateral and central amygdala differentially recruit and maintain dorsolateral striatum-dependent cocaine-seeking habits” by Jennifer E. Murray, Aude Belin-Rauscent, Marine Simon, Chiara Giuliano, Marianne Benoit-Marand, Barry J. Everitt and David Belin in Nature Communications. Published online December 14 2015 doi:10.1038/ncomms10088
Abstract
Basolateral and central amygdala differentially recruit and maintain dorsolateral striatum-dependent cocaine-seeking habits
In the development of addiction, drug seeking becomes habitual and controlled by drug-associated cues, and the neural locus of control over behaviour shifts from the ventral to the dorsolateral striatum. The neural mechanisms underlying this functional transition from recreational drug use to drug-seeking habits are unknown. Here we combined functional disconnections and electrophysiological recordings of the amygdalo-striatal networks in rats trained to seek cocaine to demonstrate that functional shifts within the striatum are driven by transitions from the basolateral (BLA) to the central (CeN) amygdala. Thus, while the recruitment of dorsolateral striatum dopamine-dependent control over cocaine seeking is triggered by the BLA, its long-term maintenance depends instead on the CeN. These data demonstrate that limbic cortical areas both tune the function of cognitive territories of the striatum and thereby underpin maladaptive cocaine-seeking habits.
“Basolateral and central amygdala differentially recruit and maintain dorsolateral striatum-dependent cocaine-seeking habits” by Jennifer E. Murray, Aude Belin-Rauscent, Marine Simon, Chiara Giuliano, Marianne Benoit-Marand, Barry J. Everitt and David Belin in Nature Communications. Published online December 14 2015 doi:10.1038/ncomms10088







