Summary: Researchers believe their finding could usher in a new era of personalized interventions to help chronic pain sufferers.
Source: Northwestern University.
New study first to pinpoint unique brain region responsible for placebo response in pain.
Scientists have identified for the first time the region in the brain responsible for the “placebo effect” in pain relief, when a fake treatment actually results in substantial reduction of pain, according to new research from Northwestern Medicine and the Rehabilitation Institute of Chicago (RIC).
Pinpointing the sweet spot of the pain killing placebo effect could result in the design of more personalized medicine for the 100 million Americans with chronic pain. The fMRI technology developed for the study has the potential to usher in an era of individualized pain therapy by enabling targeted pain medication based on how an individual’s brain responds to a drug.
The finding also will lead to more precise and accurate clinical trials for pain medications by eliminating individuals with high placebo response before trials.
The scientists discovered a unique brain region within the mid frontal gyrus that identifies placebo pill responders in one trial and can be validated (95 percent correct) in the placebo group of a second trial.
The study will be published Oct. 27, 2016, in PLOS Biology.
“Given the enormous societal toll of chronic pain, being able to predict placebo responders in a chronic pain population could both help the design of personalized medicine and enhance the success of clinical trials,” said Marwan Baliki, research scientist at RIC and an assistant professor of physical medicine and rehabilitation at Northwestern University Feinberg School of Medicine.
Baliki and Vania Apkarian, professor of physiology at Feinberg in whose lab the research was conducted, are both corresponding authors on the paper.
Using drugs to treat patients’ pain has been trial and error, with physicians changing dosage or trying another type of drug if one doesn’t work.
“The new technology will allow physicians to see what part of the brain is activated during an individual’s pain and choose the specific drug to target this spot,” Apkarian said. “It also will provide more evidence-based measurements. Physicians will be able to measure how the patient’s pain region is affected by the drug.”
Currently, placebo response is primarily studied in healthy subjects within controlled experimental settings. While such experiments aid understanding of the biological and behavioral underpinning of placebo response in experimental (applied) pain, they translate poorly to the clinic, where pain is mainly chronic in nature, Baliki said.
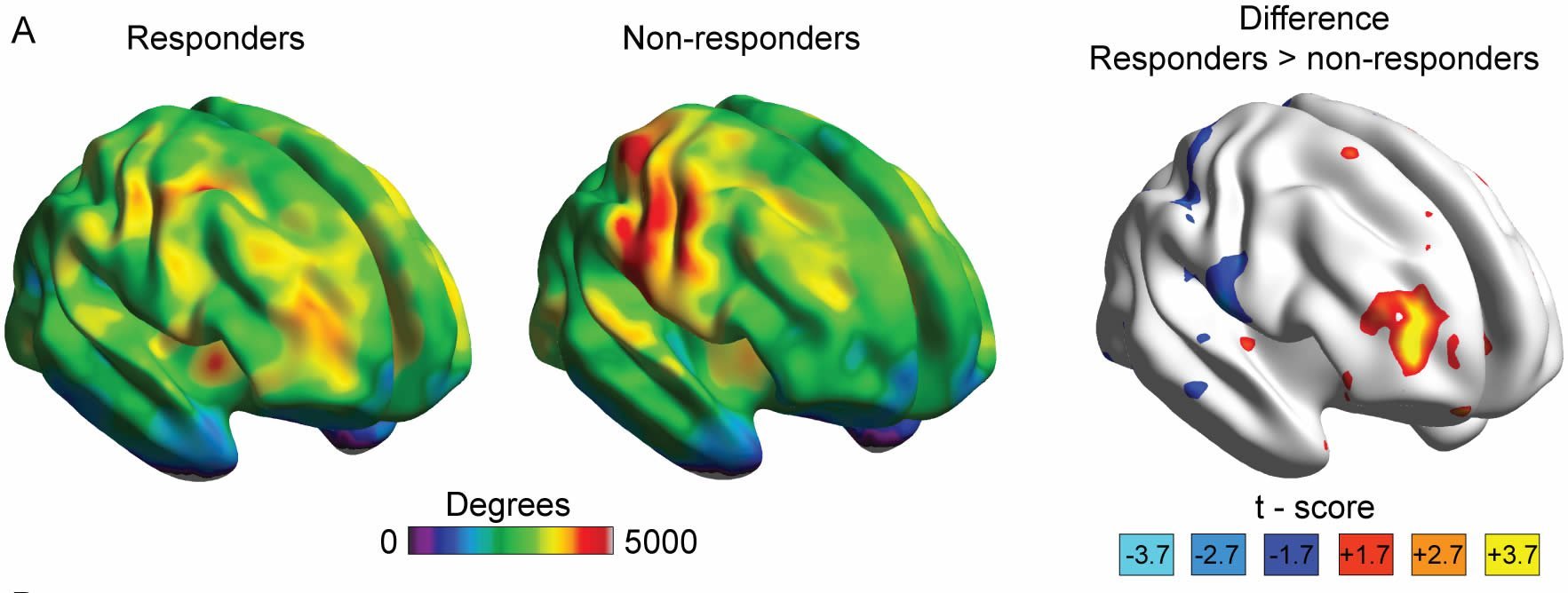
In this new study and for the first time, scientists used functional magnetic resonance imaging (fMRI) combined with a standard clinical trial design to derive an unbiased brain-based neurological marker to predict analgesia associated with placebo treatment in patients with chronic knee osteoarthritis pain. Scientists showed placebo pill ingestion is associated with a strong analgesia effect, with more than half of the patients reporting significant pain relief.
If future similar studies can further expand and eventually provide a brain-based predictive best-therapy option for individual patients, it would dramatically decrease unnecessary exposure of patients to ineffective therapies and decrease the duration and magnitude of pain suffering and opioid use, Baliki and Apkarian said.
Funding: The study was supported by grant NS035115 from the National Institute of Neurological Disorders and Stroke and grant AT007987 from the National Center for Complementary and Integrative Health, both of the National Institutes of Health. The Canadian Institutes of Health Research and Eli Lilly Pharmaceuticals also supported the research.
Source: Marla Paul – Northwestern University
Image Source: NeuroscienceNews.com image is credited to Baliki et al./PLOS Biology.
Original Research: Full open access research for “Brain Connectivity Predicts Placebo Response across Chronic Pain Clinical Trials” by Pascal Tétreault, Ali Mansour, Etienne Vachon-Presseau, Thomas J. Schnitzer, A. Vania Apkarian, and Marwan N. Baliki in PLOS Biology. Published online October 27 2016 doi:10.1371/journal.pbio.1002570
[cbtabs][cbtab title=”MLA”]Northwestern University “Placebo Sweet Spot for Pain Relief Found in Brain.” NeuroscienceNews. NeuroscienceNews, 27 October 2016.
<https://neurosciencenews.com/pain-placebo-neuroscience-5370/>.[/cbtab][cbtab title=”APA”]Northwestern University (2016, October 27). Placebo Sweet Spot for Pain Relief Found in Brain. NeuroscienceNew. Retrieved October 27, 2016 from https://neurosciencenews.com/pain-placebo-neuroscience-5370/[/cbtab][cbtab title=”Chicago”]Northwestern University “Placebo Sweet Spot for Pain Relief Found in Brain.” https://neurosciencenews.com/pain-placebo-neuroscience-5370/ (accessed October 27, 2016).[/cbtab][/cbtabs]
Abstract
Brain Connectivity Predicts Placebo Response across Chronic Pain Clinical Trials
Placebo response in the clinical trial setting is poorly understood and alleged to be driven by statistical confounds, and its biological underpinnings are questioned. Here we identified and validated that clinical placebo response is predictable from resting-state functional magnetic-resonance-imaging (fMRI) brain connectivity. This also led to discovering a brain region predicting active drug response and demonstrating the adverse effect of active drug interfering with placebo analgesia. Chronic knee osteoarthritis (OA) pain patients (n = 56) underwent pretreatment brain scans in two clinical trials. Study 1 (n = 17) was a 2-wk single-blinded placebo pill trial. Study 2 (n = 39) was a 3-mo double-blinded randomized trial comparing placebo pill to duloxetine. Study 3, which was conducted in additional knee OA pain patients (n = 42), was observational. fMRI-derived brain connectivity maps in study 1 were contrasted between placebo responders and nonresponders and compared to healthy controls (n = 20). Study 2 validated the primary biomarker and identified a brain region predicting drug response. In both studies, approximately half of the participants exhibited analgesia with placebo treatment. In study 1, right midfrontal gyrus connectivity best identified placebo responders. In study 2, the same measure identified placebo responders (95% correct) and predicted the magnitude of placebo’s effectiveness. By subtracting away linearly modeled placebo analgesia from duloxetine response, we uncovered in 6/19 participants a tendency of duloxetine enhancing predicted placebo response, while in another 6/19, we uncovered a tendency for duloxetine to diminish it. Moreover, the approach led to discovering that right parahippocampus gyrus connectivity predicts drug analgesia after correcting for modeled placebo-related analgesia. Our evidence is consistent with clinical placebo response having biological underpinnings and shows that the method can also reveal that active treatment in some patients diminishes modeled placebo-related analgesia.
“Brain Connectivity Predicts Placebo Response across Chronic Pain Clinical Trials” by Pascal Tétreault, Ali Mansour, Etienne Vachon-Presseau, Thomas J. Schnitzer, A. Vania Apkarian, and Marwan N. Baliki in PLOS Biology. Published online October 27 2016 doi:10.1371/journal.pbio.1002570







