Summary: Researchers take a look at what changes in the brain to help store information as we learn.
Source: TUE.
As we learn, something must change in our brain to store the information. Precisely what it is that is changing, and how it does so, remains a mystery. PhD student Rémy Kusters investigated what happens, at the smallest scales, in a single one of the quadrillion (a million times a billion) connections in our brain. By modeling the biophysical processes that take place there, he discovered that the shape of the connections plays a crucial role in regulating their strength. On Wednesday Oct 5, Kusters received his doctorate with honors (cum laude).
The brain contains billions of interconnected nerve cells. On each of the ‘tentacles’ of these cells, tiny ‘microspines’ reach out to the tentacles of other nerve cells. The point at which they are closest together – they never actually touch – is called the synapse. At the synapse, the sending nerve cell converts electrical signals into chemical signals, which are picked up at the receiving nerve cell and converted back into an electrical signal so that it may continue its journey. Precisely how this signal transmission is regulated at the synapse is key to our ability to learn and remember.
Using models and computer simulations, Kusters examined the physical processes that happen on, and in, the microspine. Once fully grown, dendritic spines (their official name) resemble tiny mushrooms. As it turns out, that this particular shape is important – and remarkably effective – in regulating the strength of the connection. For this strength, the number of receptors that are around to relay the signal is crucial. These receptors must, of course, come from somewhere. They are transported in small membrane containers that have to pass through the neck of the mushroom. If it is too narrow, the supply comes to a halt, weakening the strength of the brain connection.
So it would appear that the neck of the mushroom is hindering strong connections. Kusters discovered, however, that there is a different side to this story. The receptors are small proteins that are able to move around, exploring the surface of the mushroom. This means that they may spontaneously escape. Narrow necks, it turns out, make it much more difficult for the receptors to drift away. The narrow neck also serves to keep receptors already on the mushroom around for longer. So, the shape of the mushroom controls the balance between supply and loss of receptors, and thereby the strength of the neuronal connection.
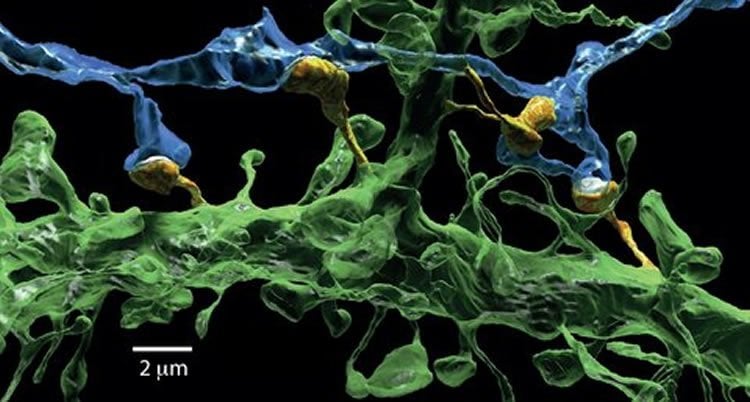
This immediately invites a deeper question – what is it that determines the shape of the spine as it matures? Kusters also studied the development of the spine shape. Spines erupt constantly, and everywhere, on a nerve cell but are not always successful in making a connection. The theoretical biophysicist’s models suggest that the protein actin, a principal component of the skeleton of most cells, facilitates the emergence of the spine and development into a mushroom. Kusters realizes that his work just a small piece in the big puzzle of how the brain works, and states that “it will still take decades before we unravel how everything works.”
Kusters’ supervisor is Kees Storm (TU/e, Applied Physics). The research is part of the FOM programme ‘Barriers in the brain: the molecular physics of learning and memory’, a collaboration between the VU (Amsterdam), the University of Leiden, the University of Utrecht and Eindhoven University of Technology.
Last Wednesday Kusters defended his thesis entitled ‘From shape to function: growth and physical regulation of dendritic spines’. He was awarded the doctorate cum laude, a rare distinction reserved for the top five percent of all PhDs awarded. Kusters will now take up a position as post-doctoral researcher at the prestigious Institut Curie in Paris.
Source: TUE
Image Source: NeuroscienceNews.com image credited to N. Kasthuri et.al/Cell.
[cbtabs][cbtab title=”MLA”]TUE. “The Secrets of the Learning Brain.” NeuroscienceNews. NeuroscienceNews, 10 October 2016.
<https://neurosciencenews.com/neuroscience-learning-neocortex-5255/>.[/cbtab][cbtab title=”APA”]TUE. (2016, October 10). The Secrets of the Learning Brain. NeuroscienceNews. Retrieved October 10, 2016 from https://neurosciencenews.com/neuroscience-learning-neocortex-5255/[/cbtab][cbtab title=”Chicago”]TUE. “The Secrets of the Learning Brain.” https://neurosciencenews.com/neuroscience-learning-neocortex-5255/ (accessed October 10, 2016).[/cbtab][/cbtabs]






