In the midst of a “crisis” in the ability to replicate the results of laboratory research, a European consortium has conducted a large-scale animal study that closely mimics a human clinical trial.
In an animal study that mimics the design and rigor of a human clinical trial, a potential stroke therapy quelled inflammation in the brains of some mice, but failed to do so in mice with a more severe form of stroke.
The results hint that the treatment, already in phase 2 human clinical trials, may potentially benefit only a subset of stroke patients, offering knowledge that could help improve design of future studies on this drug.
More broadly, the results published in the 5 August issue of Science Translational Medicine show how this type of study, called a preclinical randomized controlled trial or pRCT, may offer a potentially powerful tool for bridging the gap between the lab bench and the bedside. Designed and conducted to more stringent standards than are typical for animal studies, pRCTs could help select the most promising drug candidate before launching costly and potentially ineffective, or even harmful, clinical trials.
“Our study unequivocally proved that preclinical randomized multicenter trials are feasible, which has been doubted over the last decade,” said Gemma Llovera from the University Medical Center Munich, Germany, the study’s first author.
“We believe that pRCTs might become a new step within the pipeline from basic research [to] preclinical drug testing to clinical trials,” said senior author Arthur Liesz, also from the University Medical Center Munich.
The mouse trial was performed over the course of a year and four months and cost a total of $180,000 or €165,000, including more than €30,000 for the cost of the drug alone.
“Compared to the enormous financial and scientific loss caused by a negative clinical trial, preventing a single unnecessary or even harmful clinical trial by conducting a pRCT…will outweigh the costs and time of a pRCT manifold,” said Liesz.
Biomedical research is widely known to be plagued by the “reproducibility crisis”: the failure to replicate results of preclinical cell or animal research that normally precedes clinical or human studies.
The research community is confronted with the fact that for many fields of research, including stroke and cancer, almost all of hundreds of drug candidates successfully tested in the lab fail in clinical trials, according to Liesz.
Laboratory studies often suffer from the improper use of animal models that are poorly representative of human disease or limited sample sizes. Some studies do not randomly assign subjects to a treatment or control group, or perform “blinding” that conceals the type of treatment from the subject or researcher. Along with flawed statistical analyses or inadequate reporting of data, these issues can introduce bias or variables that make it difficult to reliably predict how humans will react to drugs tested in animal studies.
Researchers have proposed preclinical trials modeled after a clinical randomized controlled trial (RCT), the gold standard in clinical drug development, as a way to address the replication crisis. Unlike most animal studies, RCTs require often large sample sizes, randomization, proper controls such as a placebo, blinded assessment of outcomes, and stringent data analysis and reporting.
The research community is confronted with the fact that for many fields of research, including stroke and cancer, almost all of hundreds of drug candidates successfully tested in the lab fail in clinical trials, according to Liesz.
International research consortia that seek to perform pRCTs have already been established, including the European Union-funded Multicentre Preclinical Animal Research Team (Multi-PART) for stroke research and the U.S. National Institutes of Health-funded Consortium for preclinicAl assESsment of cARdioprotective therapies (CAESAR) for cardiac research.
The European consortium led by Liesz investigated a candidate drug for stroke called CD49d-specific antibody therapy, also known as natalizumab, which has been approved to treat multiple sclerosis. Four rodent studies previously showed that these antibodies protected the brain from inflammation after ischemic stroke, but these results were contradicted by a fifth study showing no benefit. Nevertheless, a phase 2 clinical trial was launched in stroke patients.
“I think most researchers in the field — including me — had the feeling that this clinical study was somewhat prematurely initiated,” said Liesz.
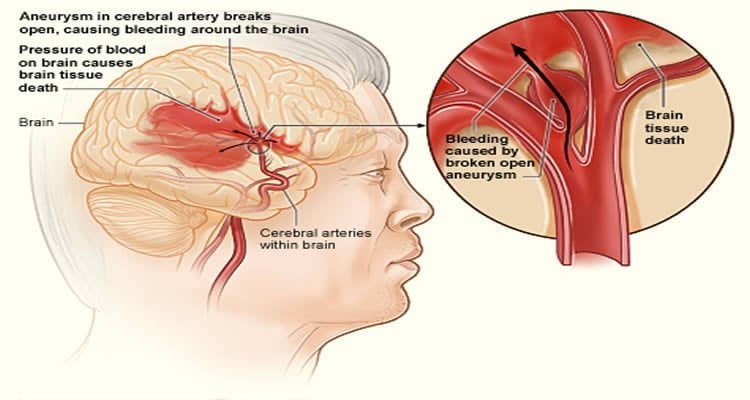
To determine whether a preclinical trial could have better guided the decision to advance the CD49d-specific antibody to clinical trials, six independent research centers in Europe collaborated to conduct a multicenter, centrally coordinated, blinded pRCT. Each lab performed experiments in two different mouse models of stroke using similar rigorous procedures and many of the same materials, down to the surgical equipment and mice of identical age and gender from the same commercial breeder. The centers pooled results from a total of 315 mice.
In mice with a form of confined stroke, the anti-CD49d antibody therapy stemmed inflammation and reduced brain damage after stroke. In another model of severe stroke, which caused more extensive brain injury, the antibody showed no protective effect in the mice, indicating that the success of the treatment may depend on the type and severity of the stroke.
“The results of our pRCT would suggest that potentially only a subpopulation of stroke patients will profit from immune-targeted stroke therapies,” said Liesz. However, he added, researchers need to better understand the inflammatory response during stroke before they can pinpoint specific patients most likely to benefit from treatment.
The researchers plan to compare their findings with the outcome of the recently completed phase 2 clinical trial, expected to be released later this year. If the preclinical and clinical results prove similar, this would support the pRCT as a valuable research tool for deciding whether or not a drug should advance to clinical testing.
“If supported by funding agencies and the pharmaceutical industry, pRCTs [may] become a mandatory step before testing a drug candidate in a clinical trial,” said Liesz.
Source: Jean Mendoza – AAAS
Image Credit: The image is in the public domain
Original Research: Abstract for “Results of a preclinical randomized controlled multicenter trial (pRCT): Anti-CD49d treatment for acute brain ischemia” by GEMMA LLOVERA, KERSTIN HOFMANN, STEFAN ROTH, ANGELICA SALAS-PÉRDOMO, MAURA FERRER-FERRER, CARLO PEREGO, ELISA R. ZANIER, UTA MAMRAK, ANDRE REX, HÉLÈNE PARTY, VÉRONIQUE AGIN, CLAUDINE FAUCHON, CYRILLE ORSET, BENOÎT HAELEWYN, MARIA-GRAZIA DE SIMONI, ULRICH DIRNAGL, ULRIKE GRITTNER, ANNA M. PLANAS, NIKOLAUS PLESNILA, DENIS VIVIEN, ARTHUR LIESZ in Science Translational Medicine. Published online August 5 2015 doi:10.1126/scitranslmed.aaa9853
Abstract
Results of a preclinical randomized controlled multicenter trial (pRCT): Anti-CD49d treatment for acute brain ischemia
Over the last few decades, clinical trial design and analysis have become increasingly stringent. These refinements—designed to ensure valid conclusions for formal drug approval—have improved clinical trial reliability. Now, Llovera et al. have applied the principles of the gold-standard randomized, controlled clinical trial to a preclinical investigation. They tested an antibody to CD49d, which inhibits leukocyte migration into the brain, in two mouse models of stroke. Data from their six-center, preclinical, randomized controlled trial in mice show that the antibody significantly reduced both leukocyte invasion and infarct volume after a small cortical stroke but that it did not have any effect in the other model, in which the animal suffered a larger injury. The authors outline the many lessons learned from their experience for further application of preclinical randomized controlled trials to translational research.
“Results of a preclinical randomized controlled multicenter trial (pRCT): Anti-CD49d treatment for acute brain ischemia” by GEMMA LLOVERA, KERSTIN HOFMANN, STEFAN ROTH, ANGELICA SALAS-PÉRDOMO, MAURA FERRER-FERRER, CARLO PEREGO, ELISA R. ZANIER, UTA MAMRAK, ANDRE REX, HÉLÈNE PARTY, VÉRONIQUE AGIN, CLAUDINE FAUCHON, CYRILLE ORSET, BENOÎT HAELEWYN, MARIA-GRAZIA DE SIMONI, ULRICH DIRNAGL, ULRIKE GRITTNER, ANNA M. PLANAS, NIKOLAUS PLESNILA, DENIS VIVIEN, ARTHUR LIESZ in Science Translational Medicine. Published online August 5 2015 doi:10.1126/scitranslmed.aaa9853