Summary: Specific inhibitory neurons in the prefrontal cortex appear to become persistently activated following acute stress. The findings may pave the way for the creation of more targeted therapeutics for stress.
Source: Vanderbilt University
Stressful experiences can lead to adaptive or detrimental behaviors. Understanding how stress can affect our brains can help understand basic brain function and is also essential to discerning causes and treatments for some diseases.
A group of researchers led by Jeffrey Conn, professor of pharmacology at Vanderbilt, explored how specific types of neurons within the prefrontal cortex, the brain area involved in decision-making, mood and motivation, responded to acute stress in models.
They found that one type of inhibitory neuron was persistently activated after acute stress, and this research implicated a receptor that has been targeted by the Warren Center for Neuroscience Drug Discovery (WCNDD) for drug development.
“We identified novel plasticity mechanisms that permit stress-induced changes to behavior. During acute stress, brain circuits encoding emotion and arousal are rapidly potentiated,” said Max Joffe, assistant professor of psychiatry at the University of Pittsburgh and former member of the Conn lab at Vanderbilt.
“Through specific inhibitory cells localized to the prefrontal cortex, this plasticity attenuates the activity of cognitive circuits that promote working memory.”
According to Conn, “the new understanding provided by these studies has direct implications for the development of potential drugs for treatment of major depression, anxiety and other disorders associated with excessive stress responses.”
Multiple factors contributed to the execution of this work. “We used a combination of specific transgenic, optogenetic and pharmacologic tools to examine cell-type specific adaptations in models,” Joffe said.
“We are deeply thankful and grateful to have had the opportunity to collaborate with many individuals within the WCNDD and the Vanderbilt Center for Addiction Research. Their efforts were instrumental in this project. We were also excited to work with the Tadross lab at Duke to employ Drugs Acutely Restricted by Tethering (DART) to selectively modulate glutamate receptors in a small group of neurons within the prefrontal cortex.”
Why it matters
In 2021, the American Psychological Association found that more than 80 percent of adults have reported emotions associated with prolonged stress. The research by Conn’s team and related efforts is important in seeking out better medications to combat conditions connected with stress and its effect on the brain.
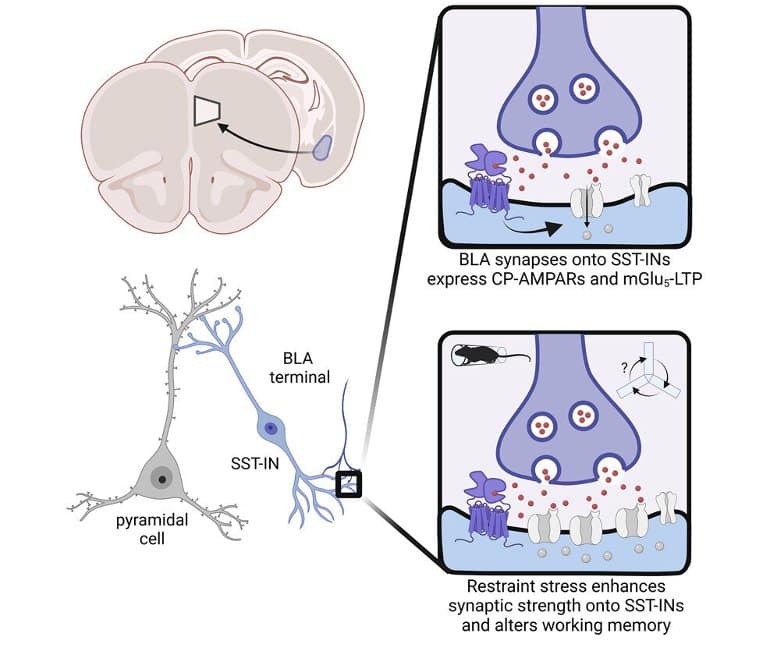
The groups’ findings increase the understanding of how stress affects brain function and how it is relevant to the cause of affective disorders, addictions and other diseases, as well as to the basic function of the brain. It may pave the way for future small-molecule medications that directly target stress.
“We hope that these results will help to renew enthusiasm for compounds that inhibit mGlu5 receptors as potential anti-stress, anxiolytic and antidepressant medications. Long term, we hope these studies will provide motivation for targeting somatostatin-expressing interneurons in the prefrontal cortex as cellular targets for new treatments for affective disorders,” Joffe said.
The researchers are now interested in understanding how the elements used to conduct this study behave in disease models, particularly chronic stress models and models of alcohol and drug use.
About this stress and neuroscience research news
Author: Press Office
Source: Vanderbilt University
Contact: Press Office – Vanderbilt University
Image: The image is credited to the researchers
Original Research: Closed access.
“Acute restraint stress redirects prefrontal cortex circuit function through mGlu5 receptor plasticity on somatostatin-expressing interneurons” by Max E. Joffe et al. Neuron
Abstract
Acute restraint stress redirects prefrontal cortex circuit function through mGlu5 receptor plasticity on somatostatin-expressing interneurons
Highlights
- Restraint stress activates PFC SST-INs
- Restraint stress enhances excitatory drive onto SST-INs and feedforward inhibition
- mGlu5 receptors regulate LTP on SST-INs
- SST-mGlu5−/− mice display unique stress-related behavioral adaptations
Summary
Inhibitory interneurons orchestrate prefrontal cortex (PFC) activity, but we have a limited understanding of the molecular and experience-dependent mechanisms that regulate synaptic plasticity across PFC microcircuit.
We discovered that mGlu5 receptor activation facilitates long-term potentiation at synapses from the basolateral amygdala (BLA) onto somatostatin-expressing interneurons (SST-INs) in mice.
This plasticity appeared to be recruited during acute restraint stress, which induced intracellular calcium mobilization within SST-INs and rapidly potentiated postsynaptic strength onto SST-INs.
Restraint stress and mGlu5 receptor activation each augmented BLA recruitment of SST-IN phasic feedforward inhibition, shunting information from other excitatory inputs, including the mediodorsal thalamus.
Finally, studies using cell-type-specific mGlu5 receptor knockout mice revealed that mGlu5 receptor function in SST-expressing cells is necessary for restraint stress-induced changes to PFC physiology and related behaviors.
These findings provide new insights into interneuron-specific synaptic plasticity mechanisms and suggest that SST-IN microcircuits may be promising targets for treating stress-induced psychiatric diseases.