“You must remember this: a kiss is just a kiss, a sigh is just a sigh.”
Contrary to the words immortalized by the piano singer in “Casablanca,” a sigh is far more than a sigh. Heaving an unconscious sigh is a life-sustaining reflex that helps preserve lung function.
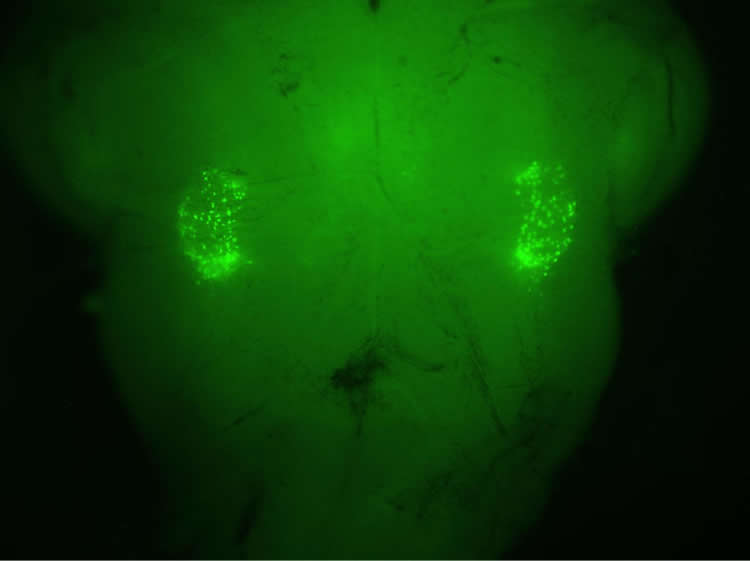
Now a new study by researchers UCLA and Stanford has pinpointed two tiny clusters of neurons in the brain stem that are responsible for transforming normal breaths into sighs. Published in the Feb. 8 advance online edition of Nature, the discovery may one day allow physicians to treat patients who cannot breathe deeply on their own — or who suffer from disorders in which frequent sighing becomes debilitating.
“Sighing appears to be regulated by the fewest number of neurons we have seen linked to a fundamental human behavior,” explained Jack Feldman, a professor of neurobiology at the David Geffen School of Medicine at UCLA and a member of the UCLA Brain Research Institute. “One of the holy grails in neuroscience is figuring out how the brain controls behavior. Our finding gives us insights into mechanisms that may underlie much more complex behaviors.”
According to Mark Krasnow, a professor of biochemistry and Howard Hughes Medical Institute Investigator at the Stanford University School of Medicine, the new findings shed light on the network of cells in the brain stem that generates breathing rhythm.
“Unlike a pacemaker that regulates only how fast we breathe, the brain’s breathing center also controls the type of breath we take,” Krasnow said. “It’s made up of small numbers of different kinds of neurons. Each functions like a button that turns on a different type of breath. One button programs regular breaths, another sighs, and the others could be for yawns, sniffs, coughs and maybe even laughs and cries.”
Using a mouse model, Krasnow and his colleagues screened more than 19,000 gene-expression patterns in the animals’ brain cells. They found roughly 200 neurons in the brain stem that manufacture and release one of two neuropeptides, which enable brain cells to talk to each other. Still, the scientists did not know which brain cells these neurons communicated with or why.
Conversely, Feldman knew that the same family of peptides, also found in humans, was highly active in a part of the brain that influences breathing and plays an important role in sighing. What he had not identified were the genes or neurons that controlled them.
By joining forces, Krasnow’s and Feldman’s labs discovered that the peptides triggered a second set of 200 neurons. These cells activate the mouse’s breathing muscles to produce a sigh — roughly 40 times an hour.
The researchers found that blocking one of the peptides cut the animals’ sighing rate in half. Silencing both peptides halted the mice’s ability to sigh completely.
“These molecular pathways are critical regulators of sighing, and define the core of a sigh-control circuit,” Krasnow said. “It may now be possible to find drugs that target these pathways to control sighing.”
Sighing is vital to lung function, and thus to life, Feldman emphasized.
“A sigh is a deep breath, but not a voluntary deep breath,” he said. “It starts out as a normal breath, but before you exhale, you take a second breath on top of it.”
On average, a person sighs every five minutes, which translates into 12 sighs per hour.
The purpose of sighing is to inflate the alveoli, the half-billion, tiny, delicate, balloon-like sacs in the lungs where oxygen enters and carbon dioxide leaves the bloodstream. Sometimes, though, individual sacs collapse.
“When alveoli collapse, they compromise the ability of the lung to exchange oxygen and carbon dioxide,” Feldman said. “The only way to pop them open again is to sigh, which brings in twice the volume of a normal breath. If you don’t sigh, your lungs will fail over time.”
Turning on sighing would be useful in people who cannot breathe deeply on their own. Early artificial breathing devices did not regularly give patients a deep breath, and many patients died. Current ventilators regularly deliver a large inflation of air that mimics a sigh.
“If you don’t sigh every five minutes of so, the alveoli will slowly collapse, causing lung failure,” Feldman said. “That’s why patients in early iron lungs had such problems, because they never sighed.”
The ability to limit the sighing reflex could prove useful in anxiety disorders and other psychiatric conditions where sighing grows debilitating.
The mechanism behind the emotional roots of conscious sighing remains a mystery.
“There is certainly a component of sighing that relates to an emotional state. When you are stressed, for example, you sigh more,” Feldman said. “It may be that neurons in the brain areas that process emotion are triggering the release of the sigh neuropeptides — but we don’t know that.”
Funding: The research was supported by the Howard Hughes Medical Institute, National Institutes of Health grants HL70029, HL40959 and NS72211, a Walter V. and Idun Berry postdoctoral fellowship, the NIH Medical Scientist Training Program, and Canadian Institutes of Health Research and Alberta Innovates Health Solutions postdoctoral fellowships.
Source: Elaine Schmidt – UCLA Health Sciences
Image Source: The image is adapted from the Radboud University press release
Original Research: Abstract for “The peptidergic control circuit for sighing” by Peng Li, Wiktor A. Janczewski, Kevin Yackle, Kaiwen Kam, Silvia Pagliardini, Mark A. Krasnow & Jack L. Feldman in Nature. Published online February 8 2016 doi:10.1038/nature16964
Abstract
The peptidergic control circuit for sighing
Sighs are long, deep breaths expressing sadness, relief or exhaustion. Sighs also occur spontaneously every few minutes to reinflate alveoli, and sighing increases under hypoxia, stress, and certain psychiatric conditions. Here we use molecular, genetic, and pharmacologic approaches to identify a peptidergic sigh control circuit in murine brain. Small neural subpopulations in a key breathing control centre, the retrotrapezoid nucleus/parafacial respiratory group (RTN/pFRG), express bombesin-like neuropeptide genes neuromedin B (Nmb) or gastrin-releasing peptide (Grp). These project to the preBötzinger Complex (preBötC), the respiratory rhythm generator, which expresses NMB and GRP receptors in overlapping subsets of ~200 neurons. Introducing either neuropeptide into preBötC or onto preBötC slices, induced sighing or in vitro sigh activity, whereas elimination or inhibition of either receptor reduced basal sighing, and inhibition of both abolished it. Ablating receptor-expressing neurons eliminated basal and hypoxia-induced sighing, but left breathing otherwise intact initially. We propose that these overlapping peptidergic pathways comprise the core of a sigh control circuit that integrates physiological and perhaps emotional input to transform normal breaths into sighs.
“The peptidergic control circuit for sighing” by Peng Li, Wiktor A. Janczewski, Kevin Yackle, Kaiwen Kam, Silvia Pagliardini, Mark A. Krasnow & Jack L. Feldman in Nature. Published online February 8 2016 doi:10.1038/nature16964