Scientists at The Scripps Research Institute (TSRI) have identified a molecule in the brain that triggers schizophrenia-like behaviors, brain changes and global gene expression in an animal model. The research gives scientists new tools for someday preventing or treating psychiatric disorders such as schizophrenia, bipolar disorder and autism.
“This new model speaks to how schizophrenia could arise before birth and identifies possible novel drug targets,” said Jerold Chun, a professor and member of the Dorris Neuroscience Center at TSRI who was senior author of the new study.
The findings were published April 7, 2014, in the journal Translational Psychiatry.
What Causes Schizophrenia?
According to the World Health Organization, more than 21 million people worldwide suffer from schizophrenia, a severe psychiatric disorder that can cause delusions and hallucinations and lead to increased risk of suicide.
Although psychiatric disorders have a genetic component, it is known that environmental factors also contribute to disease risk. There is an especially strong link between psychiatric disorders and complications during gestation or birth, such as prenatal bleeding, low oxygen or malnutrition of the mother during pregnancy.
In the new study, the researchers studied one particular known risk factor: bleeding in the brain, called fetal cerebral hemorrhage, which can occur in utero and in premature babies and can be detected via ultrasound.
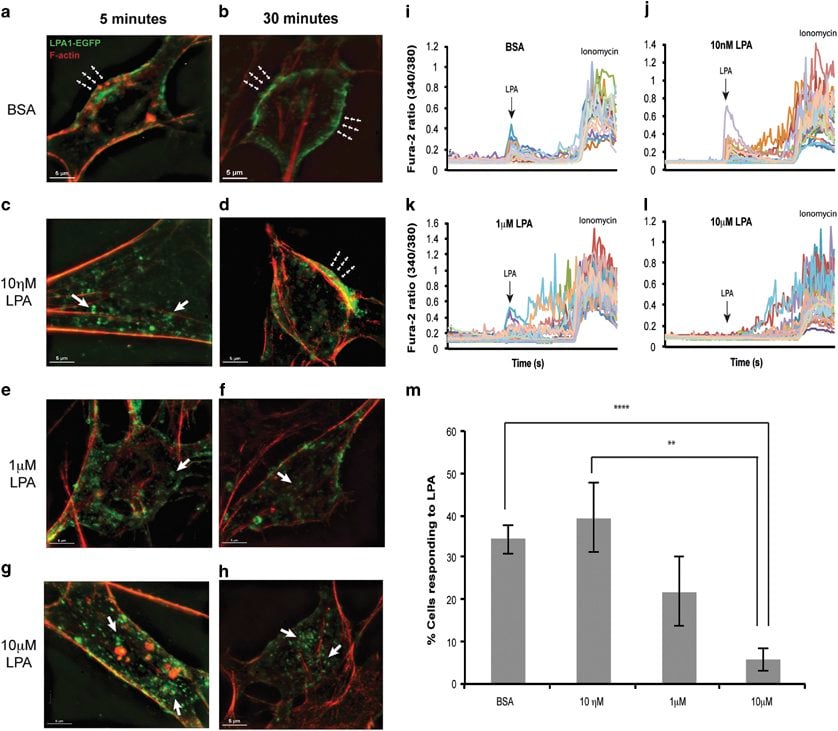
In particular, the researchers wanted to examine the role of a lipid called lysophosphatidic acid (LPA), which is produced during hemorrhaging. Previous studies had linked increased LPA signaling to alterations in architecture of the fetal brain and the initiation of hydrocephalus (an accumulation of brain fluid that distorts the brain). Both types of events can also increase the risk of psychiatric disorders.
“LPA may be the common factor,” said Beth Thomas, an associate professor at TSRI and co-author of the new study.
Mouse Models Show Symptoms
To test this theory, the research team designed an experiment to see if increased LPA signaling led to schizophrenia-like symptoms in animal models.
Hope Mirendil, an alumna of the TSRI graduate program and first author of the new study, spearheaded the effort to develop the first-ever animal model of fetal cerebral hemorrhage. In a clever experimental paradigm, fetal mice received an injection of a non-reactive saline solution, blood serum (which naturally contains LPA in addition to other molecules) or pure LPA.
Ten weeks after the mice were born, they were tested for schizophrenia-like symptoms. The researchers found that female mice given LPA-containing serum or LPA alone displayed hyperactivity upon stimulation, showed anxiety and had increased numbers of dopamine-producing neurons—all which are characteristic of schizophrenia and other psychiatric disorders.
The real litmus test to show if these symptoms were specific to psychiatric disorders, according to Mirendil, was the “prepulse inhibition test,” which measures the “startle” response to loud noises. Most mice—and humans—startle when they hear a loud noise. However, if a softer noise (known as a prepulse) is played before the loud tone, mice and humans are “primed” and startle less at the second, louder noise. Yet mice and humans with symptoms of schizophrenia startle just as much at loud noises even with a prepulse, perhaps because they lack the ability to filter sensory information.
Indeed, the female mice injected with serum or LPA alone startled regardless of whether a prepulse was placed before the loud tone.
Next, the researchers analyzed brain changes, revealing schizophrenia-like changes in neurotransmitter-expressing cells. Global gene expression studies found that the LPA-treated mice shared many similar molecular markers as those found in humans with schizophrenia. To further test the role of LPA, the researchers used a molecule to block only LPA signaling in the brain.
This treatment prevented schizophrenia-like symptoms.
Implications for Human Health
This research provides new insights, but also new questions, into the developmental origins of psychiatric disorders.
For example, the researchers only saw symptoms in female mice. Could schizophrenia be triggered by different factors in men and women as well?
“Hopefully this animal model can be further explored to tease out potential differences in the pathological triggers that lead to disease symptoms in males versus females,” said Thomas.
In addition to Chun, Thomas and Mirendil, authors of the study, “LPA signaling initiates schizophrenia-like brain and behavioral changes in a mouse model of prenatal brain hemorrhage,” were Candy De Loera of TSRI; and Kinya Okada and Yuji Inomata of the Mitsubishi Tanabe Pharma Corporation.
Funding: This research was supported by the National Institutes of Health (grants TL1 RR025772, R25 GM083275, R01 MH051699, NS084398 and R56 NS082092)
Source: The Scripps Research Institute news release
Image Source: The image was provided in the open access research paper listed below by H Mirendil, E A Thomas, C De Loera, K Okada, Y Inomata and J Chun in Translational Psychiatry (2015). Original figure is here
Research Article: Open Access Research article for “LPA signaling initiates schizophrenia-like brain and behavioral changes in a mouse model of prenatal brain hemorrhage” by H Mirendil, E A Thomas, C De Loera, K Okada, Y Inomata and J Chun in Translational Psychiatry (2015) 5, e541; Published online 7 April 2015 doi:10.1038/tp.2015.33
Additional Resources: For more information on LPA signaling, see the Chun lab’s recent review article “Lysophosphatidic Acid Signaling in the Nervous System” in the journal Neuron and “Lysophosphatidic acid signalling in development” in the journal Development.
Abstract:
From: Research article for “LPA signaling initiates schizophrenia-like brain and behavioral changes in a mouse model of prenatal brain hemorrhage” by H Mirendil, E A Thomas, C De Loera, K Okada, Y Inomata and J Chun in Translational Psychiatry (2015) 5, e541; Published online 7 April 2015 doi:10.1038/tp.2015.33
Genetic, environmental and neurodevelopmental factors are thought to underlie the onset of neuropsychiatric disorders such as schizophrenia. How these risk factors collectively contribute to pathology is unclear. Here, we present a mouse model of prenatal intracerebral hemorrhage—an identified risk factor for schizophrenia—using a serum-exposure paradigm. This model exhibits behavioral, neurochemical and schizophrenia-related gene expression alterations in adult females. Behavioral alterations in amphetamine-induced locomotion, prepulse inhibition, thigmotaxis and social interaction—in addition to increases in tyrosine hydroxylase-positive dopaminergic cells in the substantia nigra and ventral tegmental area and decreases in parvalbumin-positive cells in the prefrontal cortex—were induced upon prenatal serum exposure. Lysophosphatidic acid (LPA), a lipid component of serum, was identified as a key molecular initiator of schizophrenia-like sequelae induced by serum. Prenatal exposure to LPA alone phenocopied many of the schizophrenia-like alterations seen in the serum model, whereas pretreatment with an antagonist against the LPA receptor subtype LPA1 prevented many of the behavioral and neurochemical alterations. In addition, both prenatal serum and LPA exposure altered the expression of many genes and pathways related to schizophrenia, including the expression of Grin2b, Slc17a7 and Grid1. These findings demonstrate that aberrant LPA receptor signaling associated with fetal brain hemorrhage may contribute to the development of some neuropsychiatric disorders.






