Summary: Study reveals a new role for a scaffolding molecule that appears to play a part in glutamate receptor transportation and ultimately, memory formation.
Source: Colorado State University
Brian Falkner said, “We are our memories.”
This is one of Assistant Professor Fred Hoerndli’s favorite quotes. Hoerndli, a neuroscientist in the Department of Biomedical Sciences, and his lab are celebrating a Cell Reports publication that provides new insights into the intricacies of memory formation and maintenance.
“It took 12 years for this paper to come together and it was rewritten 60 times,” said Hoerndli. “The last part of getting it done was figuring out how to tell this very complex story.”
The brain’s ability to maintain memory can decline with age. Glutamate receptors, the most prominent signaling molecule in the brain, play a key role in encoding memory. And maintaining the number of glutamate receptors present at the synapse, or junction between two nerve cells, likely determines whether or not a memory is preserved or lost.
“It was thought for a long time that this whole process was just regulated at the synapse,” said Hoerndli. In 2009, he began more closely investigating how glutamate receptors get to synapses.
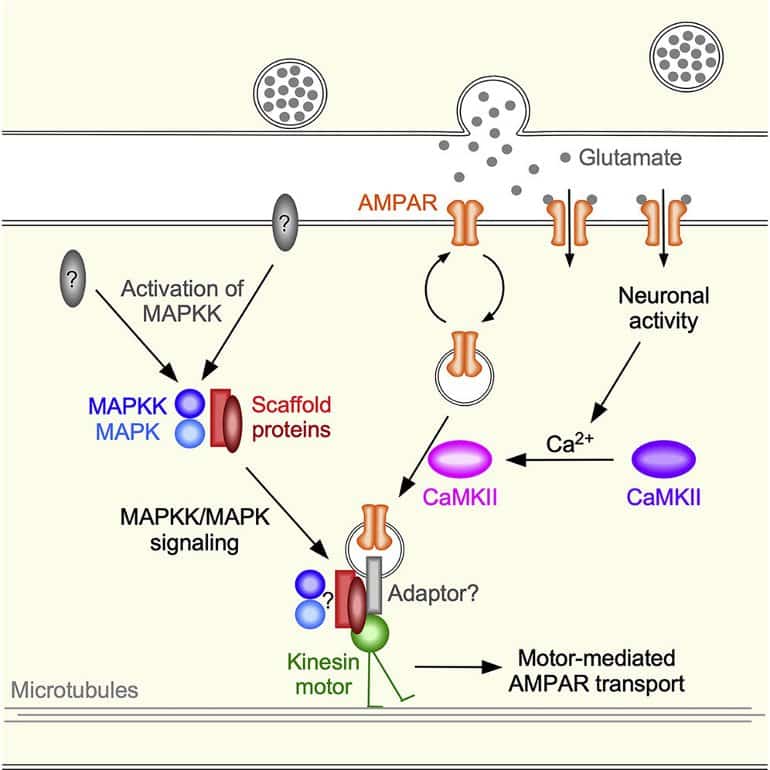
In trying to understand how this is achieved, his team uncovered a new role for a scaffolding molecule that seemed to play an unknown, until now, part in glutamate receptor transport.
By combining previous knowledge about this scaffold and what Hoerndli recently discovered about how neurons regulate transport, his team found that there were two cell signals that were happening at a synapse. And in order for transport to occur, those signals had to happen simultaneously or in close succession.
“We essentially found how two signals that have to do with memory control transport, which ultimately affects memory,” said Hoerndli. “And once we organized everything around how those two signals come together, we knew how to tell the story.”
A deceptively simple organism fuels advances in neuroscience
Hoerndli’s lab studies the cell signaling mechanisms of synaptic maintenance and memory through direct observation of C. elegans, a transparent worm with 302 neurons, a complete synaptic map, and a very short life span.
This humble worm provides labs like Hoerndli’s with an extensive library of genetic tools which allows them to dive deep into the molecular mechanisms of learning and memory.
“These studies are done in a fully alive and intact worm that has a complete nervous system, and is unharmed during the research, which allows us to see what’s going on at the molecular level,” said Hoerndli.
“It was game-changing for me as a student to learn that we could find and study genes and structures found in humans in these far simpler organisms—the fact that we can do this is mind-blowing.”
The Hoerndli lab is continuing to investigate ways various signals are controlled and work at the synapse. With greater tools and understanding of how these processes work, they hope to contribute to a more robust working body of knowledge on how to maintain memory.
About this memory research news
Author: Rhea Maze
Source: Colorado State University
Contact: Rhea Maze – Colorado State University
Image: The image is credited to the researchers
Original Research: Open access.
“MAPK signaling and a mobile scaffold complex regulate AMPA receptor transport to modulate synaptic strength” by Frédéric J. Hoerndli et al. Cell Reports
Abstract
MAPK signaling and a mobile scaffold complex regulate AMPA receptor transport to modulate synaptic strength
Highlights
- Mobile MAPK-associated scaffold proteins are required for AMPAR transport
- MAPKKs and MAPKs are required for loading scaffold proteins onto kinesin motors
- CaMKII is required for loading AMPARs onto scaffold proteins
- CaMKII and MAPK signaling contribute to the rapid exchange of synaptic AMPARs
Summary
Synaptic plasticity depends on rapid experience-dependent changes in the number of neurotransmitter receptors. Previously, we demonstrated that motor-mediated transport of AMPA receptors (AMPARs) to and from synapses is a critical determinant of synaptic strength.
Here, we describe two convergent signaling pathways that coordinate the loading of synaptic AMPARs onto scaffolds, and scaffolds onto motors, thus providing a mechanism for experience-dependent changes in synaptic strength.
We find that an evolutionarily conserved JIP-protein scaffold complex and two classes of mitogen-activated protein kinase (MAPK) proteins mediate AMPAR transport by kinesin-1 motors. Genetic analysis combined with in vivo, real-time imaging in Caenorhabditis elegans revealed that CaMKII is required for loading AMPARs onto the scaffold, and MAPK signaling is required for loading the scaffold complex onto motors.
Our data support a model where CaMKII signaling and a MAPK-signaling pathway cooperate to facilitate the rapid exchange of AMPARs required for early stages of synaptic plasticity.






