Summary: Gene therapy can restore the structure of the retina and regain normal light responses.
Source: SfN
Following gene therapy, the retina can restructure itself and regain normal light responses, according to research in mice published in Journal of Neuroscience. These results emphasize the plasticity of the retina and support the ongoing development of treatments designed to save dying cells.
Blindness is often caused by the death of rod photoreceptors, a type of cell in the retina. Current treatments have been developed that can save dying rods, but it was not known if the retina could rebuild itself after treatment, which is a key component of regaining vision.
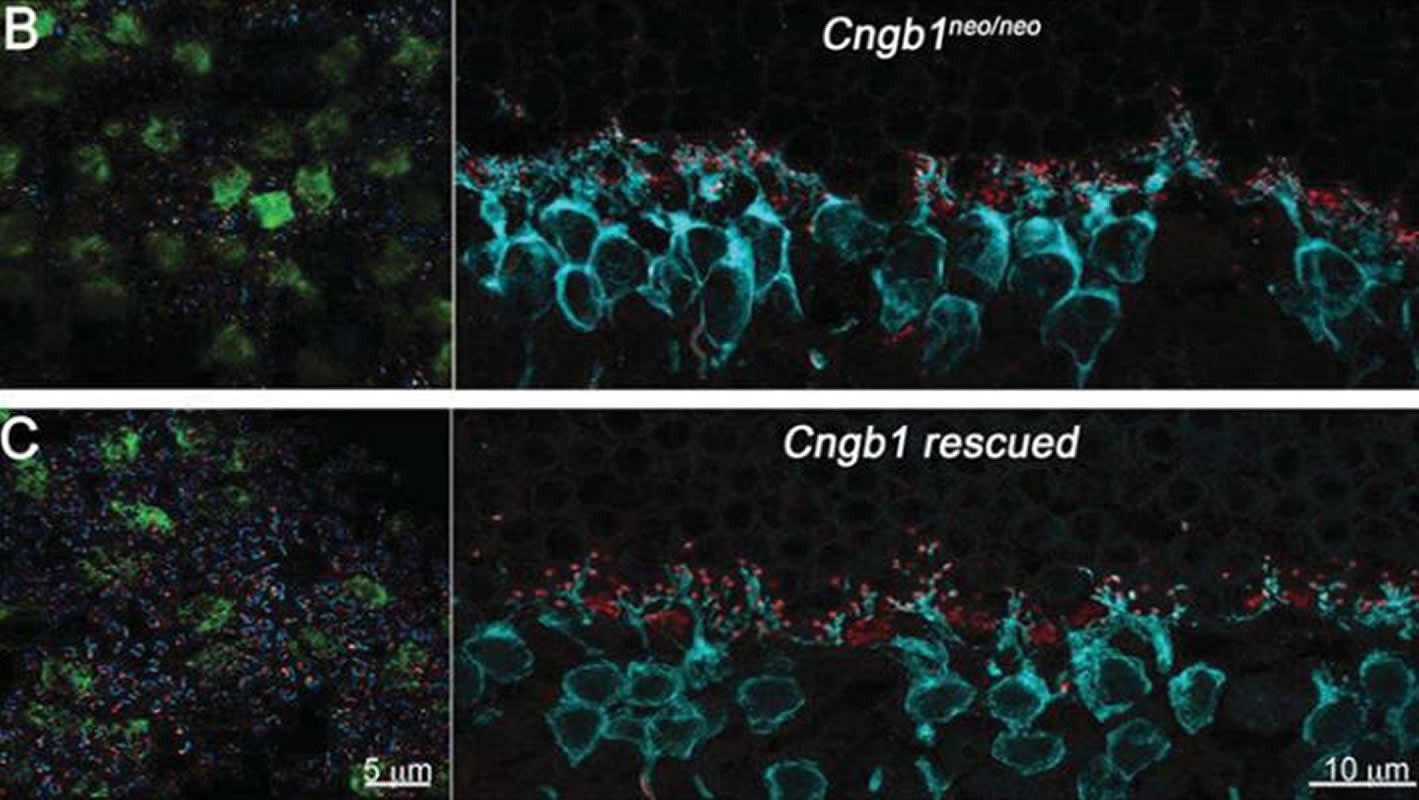
Jeannie Chen, Alapakkam Sampath, Greg Field, and colleagues developed a mouse model with genetically defective rods that mimic developmental blindness disorders in humans. Chen, Sampath, and Field’s team examined the structure of the defective retina, as well as its responses to light, at different time points with and without gene therapy. They observed that the rods that received gene therapy not only regained normal light responses, but also recovered normal connections to other retinal neurons.
Source:
SfN
Media Contacts:
Calli McMurray – SfN
Image Source:
The image is credited to Wang et al., JNeurosci 2019.
Original Research: Open access
“Activation of Rod Input in a Model of Retinal Degeneration Reverses Retinal Remodeling and Induces Formation of Functional Synapses and Recovery of Visual Signaling in the Adult Retina”. Tian Wang, Johan Pahlberg, Jon Cafaro, Rikard Frederiksen, A. J. Cooper, Alapakkam P. Sampath, Greg D. Field and Jeannie Chen.
Journal of Neuroscience. doi:10.1523/JNEUROSCI.2902-18.2019
Abstract
Activation of Rod Input in a Model of Retinal Degeneration Reverses Retinal Remodeling and Induces Formation of Functional Synapses and Recovery of Visual Signaling in the Adult Retina
A major cause of human blindness is the death of rod photoreceptors. As rods degenerate, synaptic structures between rod and rod bipolar cells disappear and the rod bipolar cells extend their dendrites and occasionally make aberrant contacts. Such changes are broadly observed in blinding disorders caused by photoreceptor cell death and are thought to occur in response to deafferentation. How the remodeled retinal circuit affects visual processing following rod rescue is not known. To address this question, we generated male and female transgenic mice wherein a disrupted cGMP-gated channel (CNG) gene can be repaired at the endogenous locus and at different stages of degeneration by tamoxifen-inducible cre-mediated recombination. In normal rods, light-induced closure of CNG channels leads to hyperpolarization of the cell, reducing neurotransmitter release at the synapse. Similarly, rods lacking CNG channel exhibit a resting membrane potential that was ∼10mV hyperpolarized compared to WT rods, indicating diminished glutamate release. Retinas from these mice undergo stereotypic retinal remodeling as a consequence of rod malfunction and degeneration. Upon tamoxifen-induced expression of CNG channels, rods recovered their structure and exhibited normal light responses. Moreover, we show that the adult mouse retina displays a surprising degree of plasticity upon activation of rod input. Wayward bipolar cell dendrites establish contact with rods to support normal synaptic transmission, which is propagated to the retinal ganglion cells. These findings demonstrate remarkable plasticity extending beyond the developmental period and support efforts to repair or replace defective rods in patients blinded by rod degeneration.
SIGNIFICANCE STATEMENT
Current strategies for treatment of neurodegenerative disorders are focused on the repair of the primary affected cell type. However, the defective neuron functions within a complex neural circuitry, which also becomes degraded during disease. It is not known whether a rescued neuron and the remodeled circuit will establish communication to regain normal function. We show that the adult mammalian neural retina exhibits a surprising degree of plasticity following rescue of rod photoreceptors. The wayward rod bipolar cell dendrites re-establish contact with rods to support normal synaptic transmission, which is propagated to the retinal ganglion cells. These findings support efforts to repair or replace defective rods in patients blinded by rod cell loss.






