Summary: The presence of amyloid-beta deposits in the retina correlated with cerebral amyloid levels in those with Alzheimer’s disease. The findings may lead to methods for the earlier detection of Alzheimer’s.
Source: UCSD
Amyloid plaques are protein deposits that collect between brain cells, hindering function and eventually leading to neuronal death. They are considered a hallmark of Alzheimer’s disease (AD), and the focus of multiple investigations designed to reduce or prevent their formation, including the nationwide A4 study.
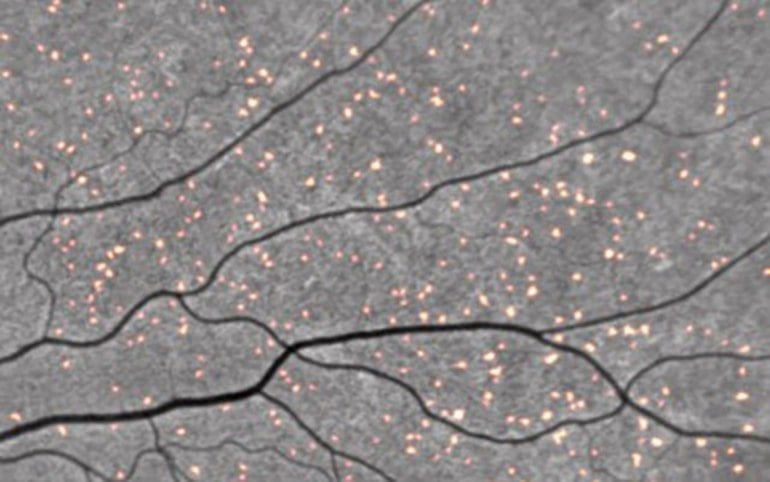
But amyloid deposits may also occur in the retina of the eye, often in patients clinically diagnosed with AD, suggesting similar pathologies in both organs.
In a small, cross-sectional study, a team of researchers, led by scientists at University of California San Diego School of Medicine, compared tests of retinal and brain amyloids in patients from the A4 study and another study (Longitudinal Evaluation of Amyloid Risk and Neurodegeneration) assessing neurodegeneration risk in persons with low levels of amyloid.
Like the proverbial “windows to the soul,” the researchers observed that the presence of retinal spots in the eyes correlated with brain scans showing higher levels of cerebral amyloid. The finding suggests that non-invasive retinal imaging may be useful as a biomarker for detecting early-stage AD risk.
The findings published in the August 17, 2021 issue of Alzheimer’s & Dementia.
“This was a small initial dataset from the screening visit. It involved eight patients,” said senior author Robert Rissman, PhD, professor of neurosciences at UC San Diego School of Medicine and director of the Biomarker Core for the Alzheimer’s Disease Cooperative Study and Alzheimer’s Disease Research Center at UC San Diego.
“But these findings are encouraging because they suggest it may be possible to determine the onset, spread and morphology of AD — a preclinical diagnosis — using retinal imaging, rather than more difficult and costly brain scans.
“We look forward to seeing the results of additional timepoint retinal scans and the impact of solanezumab (a monoclonal antibody) on retinal imaging. Unfortunately, we will need to wait to see and analyze these data when the A4 trial is completed.”
The next step, said Rissman, will be to conduct a larger study to more fully document and ascertain the relationship between retinal amyloid and cerebral amyloid, both cross-sectionally and over time.
Co-authors include: Jennifer Ngolab and Shaina Korouri, UC San Diego; Michael Donohue, Alison Belsha, Jennifer Salazar, Paula Cohen, Sandhya Jaiswal, Veasna Tan, Devon Gessert, Paul S. Aisen and Michael S. Rafii, all at University of Southern California; Neelum T. Aggarwal, Rush University Medical Center; Jessica Alber, University of Rhode Island; Ken Johnson, NeuroVision Imaging Inc; Gregory Jicha, University of Kentucky; Christopher van Dyck, Yale University; James Lah, Emory University; Stephen Salloway, Butler Hospital, R.I.; Reisa A. Sperling, Brigham and Women’s Hospital/Massachusetts General Hospital, Boston.
About this Alzheimer’s disease research news
Author: Scott La Fee
Source: UCSD
Contact: Scott La Fee – UCSD
Image: The image is credited to NeuroVision
Original Research: Open access.
“Feasibility study for detection of retinal amyloid in clinical trials: The Anti-Amyloid Treatment in Asymptomatic Alzheimer’s Disease (A4) trial” by Robert Rissman et al. Alzheimer’s and Dementia
Abstract
Feasibility study for detection of retinal amyloid in clinical trials: The Anti-Amyloid Treatment in Asymptomatic Alzheimer’s Disease (A4) trial
Introduction
The retina and brain exhibit similar pathologies in patients diagnosed with neurodegenerative diseases. The ability to access the retina through imaging techniques opens the possibility for non-invasive evaluation of Alzheimer’s disease (AD) pathology. While retinal amyloid deposits are detected in individuals clinically diagnosed with AD, studies including preclinical individuals are lacking, limiting assessment of the feasibility of retinal imaging as a biomarker for early-stage AD risk detection.
Methods
In this small cross-sectional study we compare retinal and cerebral amyloid in clinically normal individuals who screened positive for high amyloid levels through positron emission tomography (PET) from the Anti-Amyloid Treatment in Asymptomatic Alzheimer’s Disease (A4) trial as well as a companion cohort of individuals who exhibited low levels of amyloid PET in the Longitudinal Evaluation of Amyloid Risk and Neurodegeneration (LEARN) study. We quantified the number of curcumin-positive fluorescent retinal spots from a small subset of participants from both studies to determine retinal amyloid deposition at baseline.
Results
The four participants from the A4 trial showed a greater number of retinal spots compared to the four participants from the LEARN study. We observed a positive correlation between retinal spots and brain amyloid, as measured by the standardized uptake value ratio (SUVr).
Discussion
The results of this small pilot study support the use of retinal fundus imaging for detecting amyloid deposition that is correlated with brain amyloid PET SUVr. A larger sample size will be necessary to fully ascertain the relationship between amyloid PET and retinal amyloid both cross-sectionally and longitudinally.






