Summary: A new study reveals the drug QNZ-46 may help to protect patients at risk of stroke.
Source: Plymouth University.
New research shows how the novel drug QNZ-46 can help to lessen the effects of excess release of glutamate in the brain – the main cause of brain injury in stroke.
Published in Nature Communications, the study shows how identifying the source of damaging glutamate in stroke leads to discovery of brain protection with QNZ-46, a novel form of preventative treatment with clinical potential.
Existing studies show that restricted blood supply promotes the excess release of glutamate. The glutamate binds to receptors, over-stimulating them and leading to the break-down of myelin – the protective sheath around the nerve fibre (axon).
Previous studies had focused on the brain’s grey matter – the area where all of the synapses operate. Now the new study focuses on white matter – the part of the brain the connects all of the grey matter together – and demonstrates that the glutamate release from axons themselves contributes to damaging myelin.
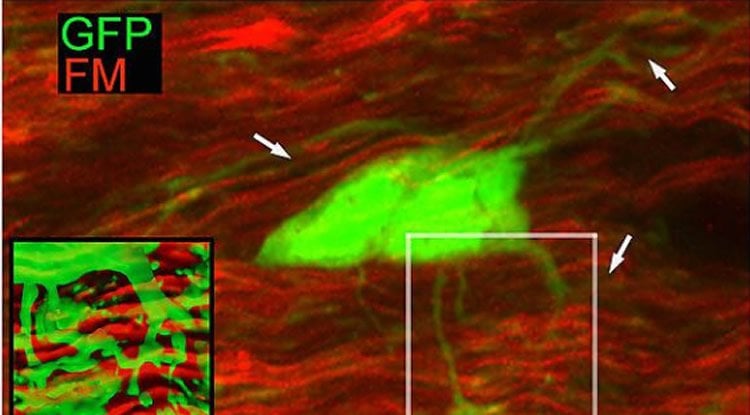
The study, led by Professor Robert Fern at the Plymouth University Peninsula Schools of Medicine and Dentistry (PUPSMD), is the first direct comparison of vesicular fusion within different cellular components in white matter, and it reveals extensive fusion in axons – a mechanism previously thought to be absent from white matter.
The findings support a rational approach toward a low-impact prophylactic therapy, such as QNZ-46, to protect patients at risk of stroke and other forms of excitotoxic injury (injury caused by excess glutamate).
As stroke is the second leading cause of disability and early death in the UK, Professor Fern, part of the University’s Institute of Translational and Stratified Medicine, explains the significance of the findings.
“Strokes are known to be caused by loss of blood to the brain, and there has been no way to treat the condition,” he said. “As much as rehabilitation can be effective, there’s nothing you can do to heal the damage. This is why it has been necessary to look at how the problem is caused in the first place.”
“Myelin damage results in severe functional deficit in the white matter of the brain, for example in ischemic – caused by lack of blood supply – stroke. By identifying how this happens, we have been able to show how QNZ-46 can be used to prevent the damage. There need to be further studies to fully understand how these findings can translate going forward, but to see that there are no negative side effects at this stage is a promising sign. By continuing further studies, we can come up with even better forms of the drug to help in stroke treatment.”
Collaborating with Professor Fern on the study were Sean Doyle, Daniel Bloch Hansen, Peter Bond, Glenn Harper from PUPSMD, and Jasmine Vella, Christian Zammit and Mario Valentino from the University of Malta.
Funding: the study was funded by the University of Plymouth and the Biotechnology and Biological Sciences Research Council.
Source: Amy McSweeny – Plymouth University
Publisher: Organized by NeuroscienceNews.com.
Image Source: NeuroscienceNews.com image is credited to the researchers.
Original Research: Open access research in Nature Communications.
doi:10.1038/s41467-018-03427-1
[cbtabs][cbtab title=”MLA”]Plymouth University “Shedding Light on the Underlying Cause of Stroke.” NeuroscienceNews. NeuroscienceNews, 15 March 2018.
<https://neurosciencenews.com/qnz-46-stroke-8640/>.[/cbtab][cbtab title=”APA”]Plymouth University (2018, March 15). Shedding Light on the Underlying Cause of Stroke. NeuroscienceNews. Retrieved March 15, 2018 from https://neurosciencenews.com/qnz-46-stroke-8640/[/cbtab][cbtab title=”Chicago”]Plymouth University “Shedding Light on the Underlying Cause of Stroke.” https://neurosciencenews.com/qnz-46-stroke-8640/ (accessed March 15, 2018).[/cbtab][/cbtabs]
Abstract
Vesicular glutamate release from central axons contributes to myelin damage
The axon myelin sheath is prone to injury associated with N-methyl-D-aspartate (NMDA)-type glutamate receptor activation but the source of glutamate in this context is unknown. Myelin damage results in permanent action potential loss and severe functional deficit in the white matter of the CNS, for example in ischemic stroke. Here, we show that in rats and mice, ischemic conditions trigger activation of myelinic NMDA receptors incorporating GluN2C/D subunits following release of axonal vesicular glutamate into the peri-axonal space under the myelin sheath. Glial sources of glutamate such as reverse transport did not contribute significantly to this phenomenon. We demonstrate selective myelin uptake and retention of a GluN2C/D NMDA receptor negative allosteric modulator that shields myelin from ischemic injury. The findings potentially support a rational approach toward a low-impact prophylactic therapy to protect patients at risk of stroke and other forms of excitotoxic injury.