Summary: Researchers have developed a new technique to quantify protein droplets associated with a range of neurodegenerative diseases, including Alzheimer’s, Parkinson’s, and ALS.
Source: Rutgers University
Some proteins in cells can separate into small droplets like oil droplets in water, but faults in this process may underlie neurodegenerative diseases in the brains of older people. Now, Rutgers researchers have developed a new method to quantify protein droplets involved in these diseases.
The novel technique, which simultaneously quantifies the surface tension and viscosity, or thickness, of protein droplets, will help scientists to study how they change, opening the way to improved understanding of the mechanisms of these diseases and the development of drug treatments.
The study appears in the journal Biophysical Reports.
The Rutgers-led team studied biomolecular condensates, which are liquid droplets that arise through the liquid-liquid phase separation of proteins and RNA inside cells in a process similar to how oil forms droplets in water.
The material properties of these protein droplets are important because they play pivotal roles in neurodegenerative diseases such as amyotrophic lateral sclerosis (ALS) and Alzheimer’s and Parkinson’s diseases. The basic idea is that liquid droplets of certain proteins can change to clogs, or aggregates of molecules, which are hallmarks of these diseases.
Surprisingly, there are no well-established methods to quantify the material properties of these protein droplets, mainly because they are very small — about a trillionth of the volume of a rain drop.
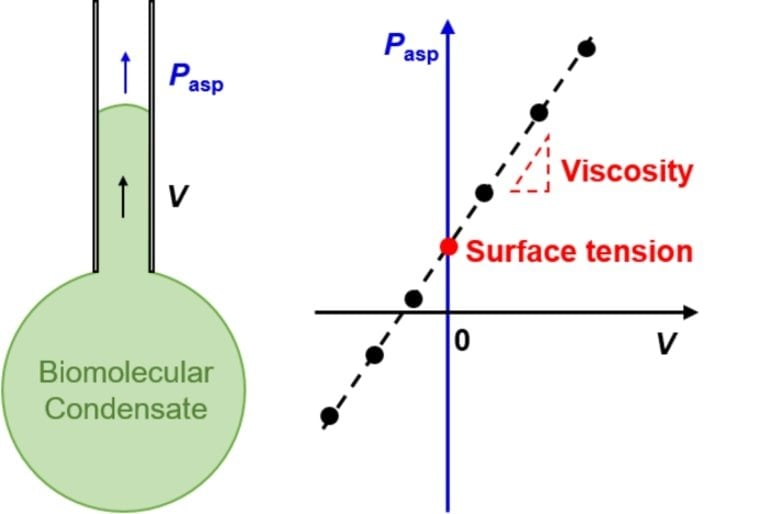
The researchers developed a straightforward method, taking inspiration from how you drink through a straw: the suction pressure in your mouth and the speed that the beverage flows in the straw can tell you the property of the liquid beverage. Similarly, one can measure the material properties of protein droplets by looking at how a droplet moves in and out of the tip of a tiny glass tube called micropipette.
The researchers looked at droplets of common liquids such as oil and water. It turns out that extreme pressure is needed to move them into a micropipette in order to overcome the high surface tension of these liquids in such a narrow passage. But once that tension is overcome, oil and water droplets move too quickly to be captured on camera because of their low viscosity. The researchers found, however, that protein droplets have just the right surface tension and viscosity to be studied quantitatively using a micropipette.
“The fact that we can apply the micropipette technique to accurately measure biomolecular condensates highlights a major difference between protein droplets and common liquids: the surface tension of protein droplets are thousands of times lower, while their viscosity are thousands of times higher than those of oil or water,” said senior author Zheng Shi, an assistant professor in the Department of Chemistry and Chemical Biology at Rutgers-New Brunswick.
“We can now finally study in a quantitative manner how material properties of protein droplets change during neurodegeneration. We anticipate this technique will be widely applicable and resolve several limitations regarding current approaches. It will open doors for unravelling the mechanisms as well as facilitating therapeutic advances in the treatment of these diseases.”
About this neuroscience research news
Author: John Cramer
Source: Rutgers University
Contact: John Cramer – Rutgers University
Image: The image is credited to Rutgers University
Original Research: Open access.
“Surface tension and viscosity of protein condensates quantified by micropipette aspiration” by Zheng Shi et al. Biophysical Reports
Abstract
Surface tension and viscosity of protein condensates quantified by micropipette aspiration
The material properties of biomolecular condensates have been suggested to play important biological and pathological roles. Despite the rapid increase in the number of biomolecules identified that undergo liquid-liquid phase separation, quantitative studies and direct measurements of the material properties of the resulting condensates have been severely lagging behind.
Here, we develop a micropipette-based technique that uniquely, to our knowledge, allows quantifications of both the surface tension and viscosity of biomolecular condensates, independent of labeling and surface-wetting effects.
We demonstrate the accuracy and versatility of this technique by measuring condensates of LAF-1 RGG domains and a polymer-based aqueous two-phase system. We further confirm our measurements using established condensate fusion and fluorescence recovery after photobleaching assays.
We anticipate the micropipette-based technique will be widely applicable to biomolecular condensates and will resolve several limitations regarding current approaches.






