Summary: Researchers have developed a new biomarker that will enable a rapid and inexpensive test for Parkinson’s disease.
Source: Kobe University
Researchers at Kobe University and Hiroshima University have successfully developed a biomarker that will enable Parkinson’s disease to be rapidly and inexpensively diagnosed from blood serum samples.
It is hoped that being able to diagnose the disease faster will also lead to the development of new treatment methods. This would be greatly beneficial, especially for aging societies like Japan.
This study was conducted by Professor Imaishi Hiromasa and Academic Researcher Ihara Kohei et al of Kobe University’s Biosignal Research Center, and Assistant Professor Oguro Ami’s research group at Hiroshima University’s Graduate School of Integrated Sciences for Life.
These research results were published in Scientific Reports on April 22, 2022.
Japan is a super-aging country. As the elderly population continues to increase, it is predicted that the number of patients suffering from neurodegenerative diseases will also rise.
Parkinson’s disease is a type of neurodegenerative disease for which a simple diagnosis method had yet to be developed. Currently, various biomarkers are in development that can be used to evaluate diseases. These biomarkers can detect the presence of a disease, as well as monitor its progress and the effectiveness of treatment.
In a world-first, the researchers developed a biomarker that can be used to easily and inexpensively assess Parkinson’s disease. It requires a mere approx. 30μ of serum from the patient. This biomarker could be used to develop diagnosis and treatment methods for Parkinson’s and advance research into discovering the molecular mechanism behind this disease.
Japan has the fastest-growing elderly population in the world and is consequently facing related issues such as the decline in elderly people’s Quality of Life (QoL) and pressure on the caregiving sector. As people age, their risk of developing various diseases increases. In particular, neurodegenerative diseases such as Parkinson’s disease have a significant impact on the sufferer’s QoL.
In addition, Parkinson’s is the second most common neurodegenerative disease in the world; believed to affect around 1~2% of the over 60s population. Furthermore, it is predicted to cost the global market 19.9 billion US dollars (including treatment costs) in 2030.
Currently, there is no way of completely curing Parkinson’s disease. Therefore, early detection is vital in order to stop its progression.
In addition, it is predicted that even earlier detection will be important in the future as various pharmaceutical companies are currently working on developing therapeutic drugs for Parkinson’s.
In light of these circumstances, there is a pressing need for a simple screening method that puts little burden on the patient. In addition, early screening methods in particular should be easy to carry out and inexpensive.
The drug metabolizing enzyme Cytochrome P450 not only metabolizes drugs but also serves as a catalyst for the oxidation of various substances. It is known that the expression of P450 inside the body changes with the onset of various diseases.
This change in P450 expression is thought to affect the quantity and quality of metabolites in the patient’s body that are related to P450 .
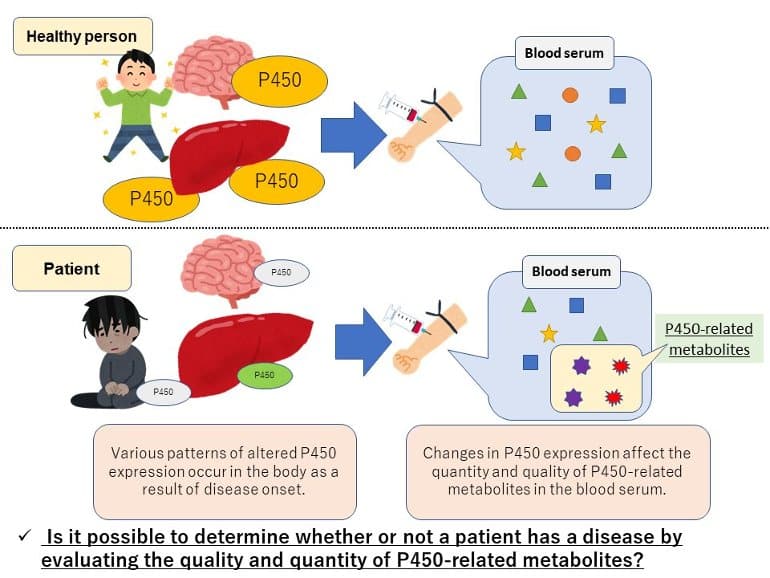
This research group previously came up with a “P450 inhibition assay” to easily detect changes in the quality and quantity of P450-related metabolites brought on by the onset of disease. This time, they became the first in the world to successfully apply this method to Parkinson’s disease diagnosis.
In the developed assay, 12 different human P450s are each mixed with a serum sample and a fluorescent substrate to cause a reaction. There are differences in quantity and quality of P450-related metabolites in the sera from healthy individuals and patients. These serum metabolites inhibit the P450-mediated oxidation of the fluorescent substrate.
By looking at the inhibition rate related to disease onset-mediated alterations in certain P450s, it is possible to discriminate sera samples from an individual with a specific disease and a healthy subject.
P450 reacts with the fluorescent substrate to generate a fluorescent substance when the assay is performed on the sera from healthy individuals. However, the reaction is different when the assay is conducted on the sera from patients and thus the obtained fluorescent values change.
In this way, the “P450 fluorescent inhibition assay” can be used to determine whether or not a disease is present by detecting these changes. It is a new liquid biopsy technique that didn’t exist until now.
The current study on Parkinson’s disease utilized the “P450 inhibition assay” that was independently developed by this research group.
Specifically, they carried out the assay on Parkinson’s disease model rats and on human patients with the disease (as well as patients with other neurodegenerative and inflammatory diseases) to evaluate whether or not the assay could be used for Parkinson’s disease diagnosis.
The results revealed that healthy individuals and individuals with Parkinson’s disease could be classified at an accuracy rate of 85~88% for both the model rat and human subjects.
The results of this study have shown that the “P450 inhibition assay” has the potential to be applied to the diagnosis of Parkinson’s disease.”
This assay only requires mere 30μ of serum and is a comparatively inexpensive testing method, therefore it is thought that it will benefit Parkinson’s disease screening.
Next, the researchers will conduct clinical performance evaluations on a larger scale, with the aim of working towards the implementation of this testing method.
About this Parkinson’s disease research news
Author: Press Office
Source: Kobe University
Contact: Press Office – Kobe University
Image: The image is in the public domain
Original Research: Open access.
“Diagnosis of Parkinson’s disease by investigating the inhibitory effect of serum components on P450 inhibition assay” by Kohei Ihara et al. Scientific Reports
Abstract
Diagnosis of Parkinson’s disease by investigating the inhibitory effect of serum components on P450 inhibition assay
Parkinson’s disease (PD) is the second most common neurodegenerative disease, and diagnostic methods and biomarkers for patients without subjective motor symptoms have not yet been established.
Previously, we developed a cytochrome P450 inhibition assay that detects alterations in metabolite levels associated with P450s caused by inflammation and exposure to endogenous or exogenous substances.
However, it is unknown whether the P450 inhibition assay can be applied in PD diagnosis. Here, we determined whether the P450 inhibition assay can discriminate sera between patients with PD and healthy individuals.
The results of the assay revealed that the P450 inhibition assay can discriminate PD with an area under the receiver operating characteristic curve (AUC) value of 0.814–0.914 in rats and an AUC value of 0.910 in humans.
These findings demonstrate that the P450 inhibition assay can aid in the future development of liquid biopsy-based diagnostic methods for PD.






