Max Planck researchers discover a new effect of oxytocin.
Sometimes small molecules are enough to alter our mood or change our metabolism. A prime example is oxytocin, which is involved in fostering emotions such as trust and love. The hormone is produced only in the brain and is released into the bloodstream by the pituitary gland. Until now it was not known why these oxytocin-producing neurons are linked to the brainstem and spinal cord. Researchers at the Max Planck Institute for Medical Research in Heidelberg have now discovered a small population of neurons that coordinate the release of oxytocin into the blood and also stimulate cells in the spinal cord. Stimulation of these cells increases oxytocin levels in the body and also has a pain-relieving effect.
Fast birth: the hormone’s name in Greek reflects one of its important functions: During childbirth, oxytocin induces contraction of the uterine muscles and initiates labour. It is also important for forming a strong bond between the mother and child and for stimulating the mother’s milk production. Furthermore, it regulates social interactions in general. It is therefore often referred to as the “cuddle hormone”.
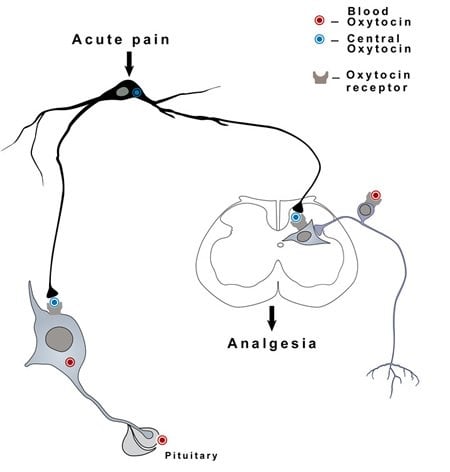
The hormone is produced only in the hypothalamus. Oxytocin-producing neurons are divided into two cell types that differ in size. The larger oxytocin-producing neurons are connected to the pituitary, which releases oxytocin into the bloodstream via capillaries. The smaller cells are connected to the brainstem and deep regions of the spinal cord. The function of these connections was unclear until now. It was suspected that they play a role in controlling the cardiovascular or respiratory system.
Small cells, big effect
Researchers at the Max Planck Institute for Medical Research and their colleagues from other countries have now discovered a pain-relieving effect of oxytocin and have determined that the hormone’s release is controlled not only via the blood but also via the spinal cord. “We’ve been able to demonstrate a new aspect of oxytocin activity and have also discovered a new subpopulation of small oxytocin-producing neurons,” director Peter Seeburg explains. “A group comprising around 30 cells of the small type extends its nerve endings to the larger neurons, which release oxytocin into the bloodstream via the pituitary and the spinal cord, where oxytocin acts as a neuron-inhibiting neurotransmitter.” This population therefore coordinates oxytocin release. “It’s fascinating that the coordination of oxytocin’s activity depends on so few cells,” Seeburg notes.
By reaching into the optogenetic toolbox, the scientists were able to stimulate the population of small cells in living experimental animals and to release more oxytocin via both pathways. Rats which then had an elevated blood oxytocin level responded less strongly to having an inflamed paw touched, indicating reduced pain sensitivity. By contrast, inhibition of the effects of oxytocin enhances pain sensitivity.
The researchers assume that the human brain also contains the same subgroup of oxytocin-producing cells. “However, the human oxytocin system is probably more complex and consists of more than thirty cells,” Seeburg explains. Moreover, the function of these cells is difficult to investigate in humans. Nevertheless, the findings could provide a new approach for the development of pain therapies.
Source: Dr. Peter H. Seeburg – Max Planck Institute
Image Credit: The images are credited to Eliava et al./Max Planck Institute.
Original Research: Abstract for “A New Population of Parvocellular Oxytocin Neurons Controlling Magnocellular Neuron Activity and Inflammatory Pain Processing” by Marina Eliava , Meggane Melchior , H. Sophie Knobloch-Bollmann, Jérôme Wahis, Miriam da Silva Gouveia, Yan Tang , Alexandru Cristian, Ciobanu , Rodrigo Triana del Rio, Lena C. Roth , Ferdinand Althammer,Virginie Chavant , Yannick Goumon , Tim Gruber, Nathalie Petit-Demoulière, Marta Busnelli, Bice Chini , Linette L. Tan, Mariela Mitre, Robert C. Froemke, Moses V. Chao, Günter Giese , Rolf Sprengel , Rohini Kuner , Pierrick Poisbeau , Peter H. Seeburg , Ron Stoop , Alexandre Charlet and Valery Grinevich in Neuron. Published online January 22 2016 doi:10.1016/j.neuron.2016.01.041
Abstract
A New Population of Parvocellular Oxytocin Neurons Controlling Magnocellular Neuron Activity and Inflammatory Pain Processing
Highlights
•Thirty parvocellular oxytocin neurons (ParvOT) alleviate acute pain
•ParvOT project to WDR sensory neurons in spinal cord (SC)
•ParvOT activate OT release from magnocellular OT neurons (magnOT)
•Dual pain suppression by peripheral magnOT and central SC OT
Summary
Oxytocin (OT) is a neuropeptide elaborated by the hypothalamic paraventricular (PVN) and supraoptic (SON) nuclei. Magnocellular OT neurons of these nuclei innervate numerous forebrain regions and release OT into the blood from the posterior pituitary. The PVN also harbors parvocellular OT cells that project to the brainstem and spinal cord, but their function has not been directly assessed. Here, we identified a subset of approximately 30 parvocellular OT neurons, with collateral projections onto magnocellular OT neurons and neurons of deep layers of the spinal cord. Evoked OT release from these OT neurons suppresses nociception and promotes analgesia in an animal model of inflammatory pain. Our findings identify a new population of OT neurons that modulates nociception in a two tier process: (1) directly by release of OT from axons onto sensory spinal cord neurons and inhibiting their activity and (2) indirectly by stimulating OT release from SON neurons into the periphery.
“A New Population of Parvocellular Oxytocin Neurons Controlling Magnocellular Neuron Activity and Inflammatory Pain Processing” by Marina Eliava , Meggane Melchior , H. Sophie Knobloch-Bollmann, Jérôme Wahis, Miriam da Silva Gouveia, Yan Tang , Alexandru Cristian, Ciobanu , Rodrigo Triana del Rio, Lena C. Roth , Ferdinand Althammer,Virginie Chavant , Yannick Goumon , Tim Gruber, Nathalie Petit-Demoulière, Marta Busnelli, Bice Chini , Linette L. Tan, Mariela Mitre, Robert C. Froemke, Moses V. Chao, Günter Giese , Rolf Sprengel , Rohini Kuner , Pierrick Poisbeau , Peter H. Seeburg , Ron Stoop , Alexandre Charlet and Valery Grinevich in Neuron. Published online January 22 2016 doi:10.1016/j.neuron.2016.01.041






