New imaging technique shows how cocaine shuts down blood flow in mouse brains.
A new method for measuring and imaging how quickly blood flows in the brain could help doctors and researchers better understand how drug abuse affects the brain, which may aid in improving brain-cancer surgery and tissue engineering, and lead to better treatment options for recovering drug addicts. The new method, developed by a team of researchers from Stony Brook University in New York, USA and the U.S. National Institutes of Health, was published today in The Optical Society’s (OSA) open-access journal Biomedical Optics Express.
The researchers demonstrated their technique by using a laser-based method of measuring how cocaine disrupts blood flow in the brains of mice. The resulting images are the first of their kind that directly and clearly document such effects, according to co-author Yingtian Pan, associate professor in the Department of Biomedical Engineering at Stony Brook University. “We show that quantitative flow imaging can provide a lot of useful physiological and functional information that we haven’t had access to before,” he says.
Drugs such as cocaine can cause aneurysm-like bleeding and strokes, but the exact details of what happens to the brain’s blood vessels have remained elusive—partly because current imaging tools are limited in what they can see, Pan says. But using their new and improved methods, the team was able to observe exactly how cocaine affects the tiny blood vessels in a mouse’s brain. The images reveal that after 30 days of chronic cocaine injection or even after just repeated acute injection of cocaine, there’s a dramatic drop in blood flow speed. The researchers were, for the first time, able to identify cocaine-induced microischemia, when blood flow is shut down—a precursor to a stroke.
Measuring blood flow is crucial for understanding how the brain is working, whether you’re a brain surgeon or a neuroscientist studying how drugs or disease influence brain physiology, metabolism and function, Pan said. Techniques like functional magnetic resonance imaging (fMRI) provide a good overall map of the flow of deoxygenated blood, but they don’t have a high enough resolution to study what happens inside tiny blood vessels called capillaries. Meanwhile, other methods like two-photon microscopy, which tracks the movement of red blood cells labeled with fluorescent dyes, have a small field of view that only measures few vessels at a time rather than blood flow in the cerebrovascular networks.
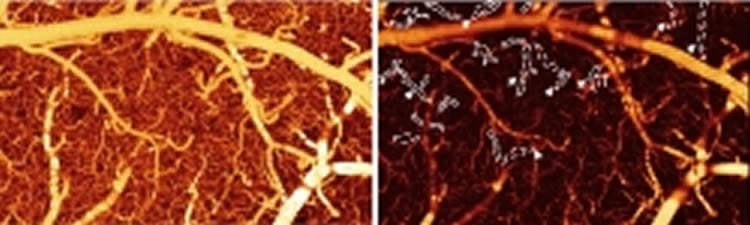
In the last few years, researchers including Pan and his colleagues have developed another method called optical coherence Doppler tomography (ODT). In this technique, laser light hits the moving blood cells and bounces back. By measuring the shift in the reflected light’s frequency—the same Doppler effect that causes the rise or fall of a siren’s pitch as it moves toward or away from you—researchers can determine how fast the blood is flowing.
It turns out that ODT offers a wide field of view at high resolution. “To my knowledge, this is a unique technology that can do both,” Pan said. And, it doesn’t require fluorescent dyes, which can trigger harmful side effects in human patients or leave unwanted artifacts—from interactions with a drug being tested, for example—when used for imaging animal brains.
Two problems with conventional ODT right now, however, are that it’s only sensitive to a limited range in blood-flow speeds and not sensitive enough to detect slow capillary flows, Pan explained. The researchers’ new method described in today’s Biomedical Optics Express paper incorporates a new processing method called phase summation that extends the range and allows for imaging capillary flows.
Another limitation of conventional ODT is that it doesn’t work when the blood vessel is perpendicular to the incoming laser beam. In an image, the part of the vessel that’s perpendicular to the line of sight wouldn’t be visible, instead appearing dark. But by tracking the blood vessel as it slopes up or down near this dark spot, the researchers developed a way to use that information to interpolate the missing data more accurately.
ODT can only see down to 1-1.5 millimeters below the surface, so the method is limited to smaller animals if researchers want to probe into deeper parts of the brain. But, Pan says, it would still be useful when the brain’s exposed in the operating room, to help surgeons operate on tumors, for example.
The new method is best suited to look at small blood vessels and networks, so it can be used to image the capillaries in the eye as well. Bioengineers can also use it to monitor the growth of new blood vessels when engineering tissue, Pan said. Additionally, information about blood flow in the brain could also be applied to developing new treatment options for recovering drug addicts.
Contact: Angela Stark – The Optical Society
Source: The Optical Society press release
Image Source: The image is credited to Biomedical Optics Express and is adapted from the Optical Society press release
Original Research: Full open access research for “Optical coherence Doppler tomography for quantitative cerebral blood flow imaging” by Jiang You, Congwu Du, Nora D. Volkow, and Yingtian Pan in Biomedical Optics Express. Published online August 2014 doi:10.1364/BOE.5.003217
Optical coherence Doppler tomography for quantitative cerebral blood flow imaging
Optical coherence Doppler tomography (ODT) is a promising neurotechnique that permits 3D imaging of the cerebral blood flow (CBF) network; however, quantitative CBF velocity (CBFv) imaging remains challenging. Here we present a simple phase summation method to enhance slow capillary flow detection sensitivity without sacrificing dynamic range for fast flow and vessel tracking to improve angle correction for absolute CBFv quantification. Flow phantom validation indicated that the CBFv quantification accuracy increased from 15% to 91% and the coefficient of variation (CV) decreased 9.3-fold; in vivo mouse brain validation showed that CV decreased 4.4-/10.8- fold for venular/arteriolar flows. ODT was able to identify cocaine-elicited microischemia and quantify CBFv disruption in branch vessels and capillaries that otherwise would have not been possible.
“Optical coherence Doppler tomography for quantitative cerebral blood flow imaging” by Jiang You, Congwu Du, Nora D. Volkow, and Yingtian Pan in Biomedical Optics Express, August 2014 doi:10.1364/BOE.5.003217