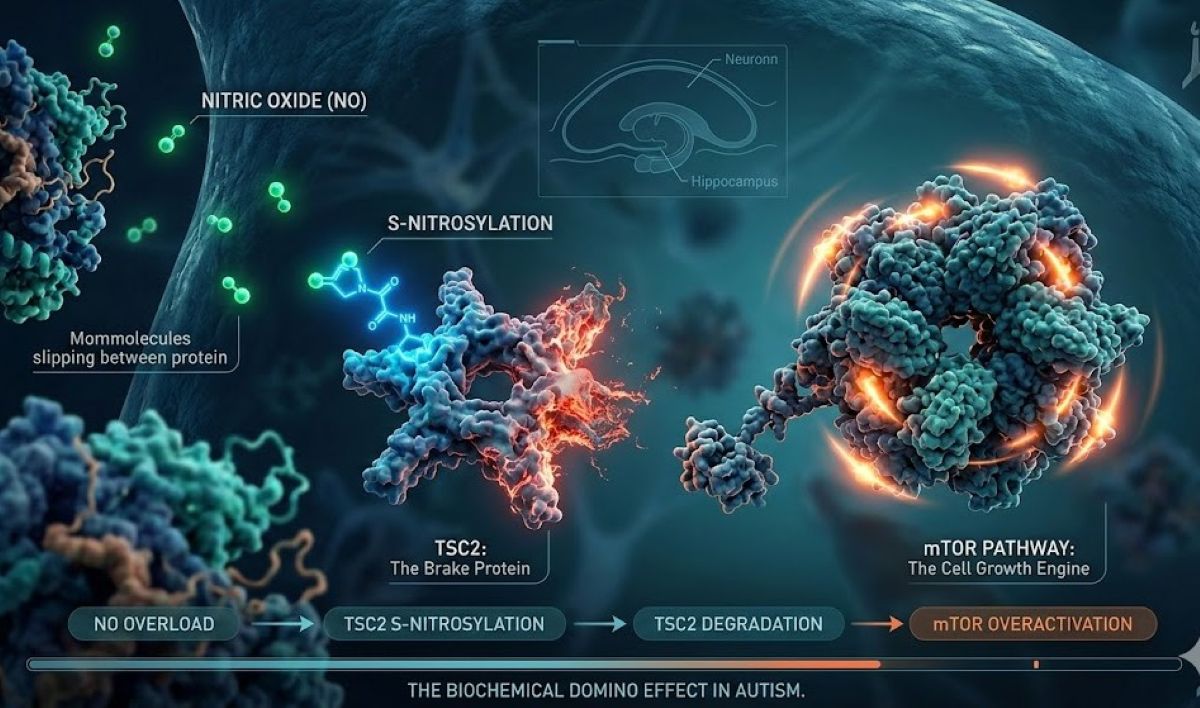
Summary: Nitric oxide (NO) is normally a subtle messenger in the brain, but new research reveals it may play a much more aggressive role in autism spectrum disorder (ASD). The study outlines a “biochemical domino effect”: excessive nitric oxide attaches to a protective protein called TSC2 through a process called S-nitrosylation.
This chemical “tag” marks the TSC2 protein for destruction. Since TSC2 normally acts as the “brake” for the mTOR pathway (a master regulator of cell growth and protein production), its removal sends mTOR into overdrive. This signaling imbalance disrupts how neurons communicate, providing a clear molecular “map” for how diverse autism risk factors converge on a single cellular pathway.
Key Facts
- The “Brake” Failure: TSC2 is a critical checkpoint. When nitric oxide levels are too high, the “brakes” on protein production are removed, leading to the cellular dysregulation often seen in ASD.
- Targeted Resistance: Researchers engineered a version of TSC2 that nitric oxide could not modify. This “resistant” protein successfully normalized mTOR signaling, proving the chemical modification is a primary driver of the pathology.
- Clinical Validation: The team found the same molecular patterns—reduced TSC2 and hyperactive mTOR—in clinical samples from children with both SHANK3 mutations and idiopathic (unknown cause) autism.
- Therapeutic Hope: Using nitric oxide inhibitors prevented the TSC2 modification and restored normal cellular function, suggesting a new class of drugs for ASD.
Source: Hebrew University of Jerusalem
Nitric oxide is usually one of the brain’s quiet helpers: a tiny molecule that slips between cells, fine-tuning communication and keeping neural systems responsive.
But new research from the Hebrew University of Jerusalem, led by Prof. Haitham Amal, The Satell Family Professor of Brain Sciences, suggests that in some forms of autism spectrum disorder (ASD), nitric oxide may also help set off a biochemical domino effect—one that pushes a key cellular control system into overdrive.
The study was first-authored by PhD student Shashank Ojha, published in Molecular Psychiatry, explores a molecular pathway that connects three important players: nitric oxide, a protective protein called TSC2, and the mTOR pathway, a major regulator of how cells grow and produce proteins.
Many researchers have suspected that mTOR signaling can become dysregulated in ASD. What has been harder to pin down is the “how”, the specific steps that might link risk factors to mTOR changes in the brain.
Prof. Amal’s team focused on a process called S-nitrosylation, a chemical modification that occurs when nitric oxide attaches to proteins and changes their behavior. Using a systems-level protein analysis approach, the researchers found that proteins involved in the mTOR pathway were especially affected, prompting them to investigate a critical checkpoint: TSC2, which normally acts like a brake on mTOR activity.
Their experiments indicated that nitric oxide can modify TSC2 in a way that marks it for removal. With less TSC2 available, the brake weakens and mTOR activity can rise. Because mTOR influences protein production and other essential cellular functions, this kind of overactivation may affect how neurons function and communicate.
To test whether this pathway could be interrupted, the team used pharmacological approaches that reduce nitric oxide production in neurons. When nitric oxide signaling was dampened, the researchers observed prevention of the TSC2 modification and a normalization of mTOR activity, along with improvements in measures linked to altered protein translation and autism-related outcomes in their experimental system.
In a complementary approach, the researchers engineered a version of TSC2 designed to resist nitric oxide-related modification. Preventing that single chemical “tag” helped protect TSC2 levels and reduced downstream effects tied to excessive mTOR signaling, supporting the idea that this specific modification may play a meaningful role in driving the pathway.
Importantly, the study also examined clinical samples from children with ASD, including children with SHANK3 mutations as well as idiopathic ASD (cases without a single known genetic cause) recruited by Dr. Adi Aran, MD.
The researchers reported patterns consistent with the proposed mechanism, including reduced TSC2 levels and increased mTOR signaling activity, which adds real-world relevance to the molecular findings.
“Autism is not one condition with one cause, and we don’t expect one pathway to explain every case,” said Prof. Haitham Amal.
“But by identifying a clearer chain of events, how nitric oxide-related changes can affect a key regulator like TSC2 and, in turn, mTOR, we hope to provide a more precise map for future research and, eventually, more targeted therapeutic ideas.”
This study further emphasizes the importance of developing nitric oxide inhibitors for ASD. Further, by outlining a specific nitric oxide–TSC2–mTOR connection, the study offers a fresh framework for understanding how cellular signaling can go off-balance in ASD and suggests new places scientists can look when developing and testing future interventions.
About Autism Spectrum Disorder (ASD)
ASD is a neurodevelopmental condition characterized by differences in social communication and behavior. It is highly diverse, with many genetic and biological factors contributing to risk and outcomes.
Researchers increasingly study cellular pathways like mTOR because they influence how brain cells grow, adapt, and build connections.
Key Questions Answered:
A: Not at all! Nitric oxide is essential for healthy brain function. The problem in ASD is a “Goldilocks” issue—too much of a good thing. When nitric oxide levels spike, it starts “tagging” proteins like TSC2 that it should normally leave alone, causing a cellular traffic jam.
A: While autism has many causes, many of them might lead to this same “clogged pipe” in the mTOR pathway. Whether the cause is genetic or environmental, if it results in too much nitric oxide, the end result on the brain’s “brakes” (TSC2) is the same.
A: While not a standard diagnostic tool yet, this study showed consistent patterns in children with ASD. Future research could lead to biomarkers that check for these specific chemical “tags” (S-nitrosylation) to help identify who might benefit most from nitric oxide-targeted therapies.
Editorial Notes:
- This article was edited by a Neuroscience News editor.
- Journal paper reviewed in full.
- Additional context added by our staff.
About this Autism research news
Author: Danae Marx
Source: Hebrew University of Jerusalem
Contact: Danae Marx – Hebrew University of Jerusalem
Image: The image is credited to Neuroscience News
Original Research: Open access.
“Nitric Oxide-Mediated S-Nitrosylation of TSC2 Drives mTOR dysregulation across Shank3 and Cntnap2 Models of Autism Spectrum Disorder” by Shashank Kumar Ojha, Maryam Kartawy, Wajeha Hamoudi, Manish Kumar Tripathi, Adi Aran & Haitham Amal. Molecular Psychiatry
DOI:10.1038/s41380-026-03514-6
Abstract
Nitric Oxide-Mediated S-Nitrosylation of TSC2 Drives mTOR dysregulation across Shank3 and Cntnap2 Models of Autism Spectrum Disorder
Autism spectrum disorder (ASD) is a complex neurodevelopmental disorder characterized by core behavioral symptoms. We previously showed that nitric oxide (NO) plays a key role in ASD.
However, the precise molecular mechanism through which NO acts via its posttranslational modification, S-nitrosylation (SNO), in ASD remains largely unknown.
Emerging evidence, including our previous studies, suggests that the mechanistic target of the rapamycin (mTOR) signaling pathway plays a critical role in ASD pathophysiology. Our SNO-proteome systems biology analysis showed the enrichment of the mTOR pathway.
In this study, we deciphered a novel mechanism of the cross talk between NO and mTOR pathway using two well-established mouse models as well as clinical samples of children with ASD.
To assess changes in the SNO-proteome, we used the SNOTRAP method, revealing increased S-nitrosylation of tuberous sclerosis complex 2 (TSC2) in Shank3Δ4–22 and Cntnap2(-/-) mutant mice.
We proved that this modification led to the loss of TSC2 protein via ubiquitination, resulting in dysregulated mTOR signaling in both excitatory and inhibitory neurons. Pharmacological inhibition of neuronal nitric oxide synthase (nNOS) successfully prevented TSC2 S-nitrosylation, mTOR overactivation, and altered protein translation in ASD models, highlighting NO’s role in modulating mTOR function.
To further validate the role of TSC2 S-nitrosylation in ASD, we generated a cysteine-to-serine mutation (C203S) in TSC2 to prevent its S-nitrosylation. Intracranial injection of the mutant TSC2 (C203S) in Shank3Δ4–22 mice in the prefrontal cortex prevented ASD-like behaviors, confirming the pathogenic role of NO-mediated TSC2 modification.
Critically, analysis of clinical samples from children with ASD, including those with SHANK3 mutations and idiopathic ASD, revealed reduced TSC2 levels and increased mTOR signaling activity, further validating our findings.
Collectively, this study uncovers a novel molecular mechanism by which S-nitrosylation disrupts TSC2 function, leading to aberrant mTOR signaling and ASD-like phenotypes.
By revealing a unique SNO-TSC2-mTOR axis, our work deciphers the novel nitric oxide-mediated mTOR activation and opens new avenues for targeted therapeutic strategies in ASD, including those carrying SHANK3 mutations.