Summary: Removing new neurons generates as a result of neurogenesis following brain injury reduced seizure activity in mouse models of mesial temporal lobe epilepsy.
Source: SfN
Removing new neurons born after a brain injury reduces seizures in mice, according to new research in Journal of Neuroscience. This approach could potentially help prevent post-injury epilepsy.
New neurons generated following a brain injury often do not develop normally. Left untreated, these cells may contribute to the development of epilepsy.
Jenny Hsieh and colleagues at the University of Texas at San Antonio continually removed new neurons that formed during the eight weeks following a seizure in mice. Hsieh’s team monitored seizure activity in the mice and observed that the treated mice experienced a 65 percent reduction in seizures compared to the untreated mice. This effect required more than four weeks of continuous treatment.
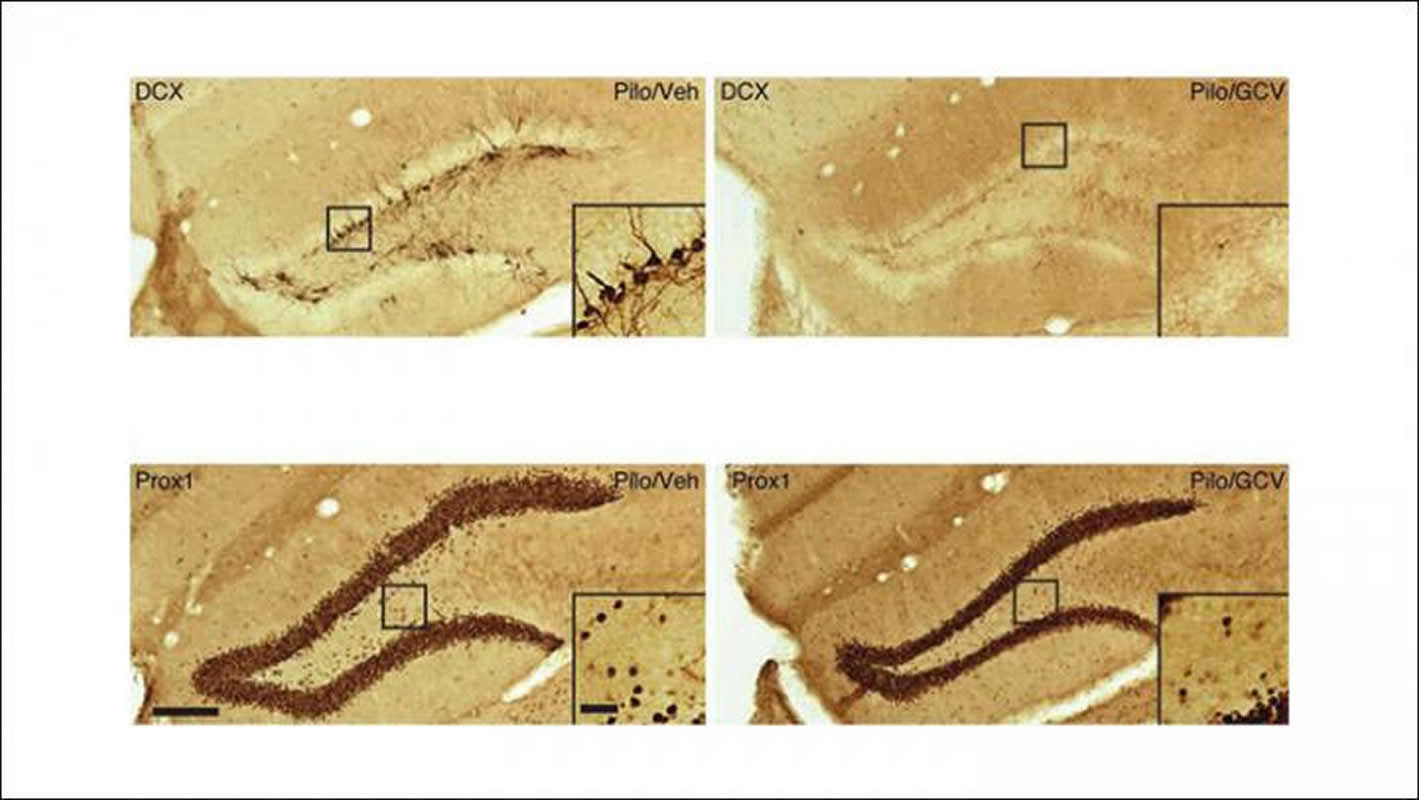
Although these findings support a role for newborn neurons in epilepsy development, they also suggest additional factors are involved. Further research may bring us closer to complete prevention of injury-induced epilepsy.
Source:
SfN
Media Contacts:
Calli McMurray – SfN
Image Source:
The image is credited to Varma et al., JNeurosci 2019.
Original Research: Closed access
“Targeting seizure-induced neurogenesis in a clinically-relevant time-period leads to transient but not persistent seizure reduction”. Parul Varma, Rebecca Brulet, Ling Zhang and Jenny Hsieh.
Journal of Neuroscience. doi:10.1523/JNEUROSCI.0920-19.2019
Abstract
Targeting seizure-induced neurogenesis in a clinically-relevant time-period leads to transient but not persistent seizure reduction
Mesial temporal lobe epilepsy (mTLE), the most common form of medically refractory epilepsy in adults is usually associated with hippocampal pathophysiology. Using rodent models of mTLE, many studies including work from our laboratory have shown that new neurons born around the onset of severe acute seizures known as status epilepticus (SE) are crucial for the process of epileptogenesis and targeting seizure-induced neurogenesis either genetically or pharmacologically can impact the frequency of chronic seizures. However, these studies are limited in their clinical relevance as none of them determine the potential of blocking new neurons generated after the epileptogenic insult to alleviate the development of chronic seizures. Therefore, using a pilocarpine-induced SE model of mTLE in mice of either sex, we show that greater than 4 weeks of continuous and concurrent ablation of seizure-induced neurogenesis after SE can reduce the formation of spontaneous recurrent seizures (SRS) by 65%. We also found that blocking post-SE neurogenesis does not lead to long-term seizure reduction as the effect was observed only transiently for 10 days with more than 4 weeks of continuous and concurrent ablation of seizure-induced neurogenesis. Thus, these findings provide evidence that seizure-induced neurogenesis when adequately reduced in a clinically relevant time-period has the potential to transiently suppress recurrent seizures, but additional mechanisms need to be targeted to permanently prevent epilepsy development.
SIGNIFICANCE STATEMENT
Consistent with morphological and electrophysiological studies suggesting aberrant adult-generated neurons contribute to epilepsy development, ablation of seizure induced new neurons at the time of the initial insult reduces the frequency of recurrent seizures. In this study, we show that continuous targeting of post-insult new neurons in a therapeutically relevant time period reduces chronic seizures, however, this effect does not persist suggesting possible additional mechanisms.