Summary: Using monoclonal antibodies instead of conventional immunosuppressant drugs preserves stem cells in mouse brains.
Source: University of Michigan
A new approach to stem cell therapy that uses antibodies instead of traditional immunosuppressant drugs robustly preserves cells in mouse brains and has potential to fast-track trials in humans, a Michigan Medicine study suggests.
For this study, researchers used monoclonal antibodies to suppress the immune system in mice and compared the results to traditional immunosuppression with the medications tacrolimus and mycophenolate mofetil. They tracked implanted human neural stem cell survival using luciferase, the protein that makes fireflies glow.
Results published in Clinical and Translational Medicine reveal that suppression with monoclonal antibodies enabled long-term survival of human stem cell transplants in mouse brains for at least six to eight months, while the cell grafts did not survive more than two weeks in most animals when using standard immunosuppressant drugs.
“This study makes it clear that using monoclonal antibodies is better for the study of stem cell transplants in the brain and spinal cord over the long term,” said lead author Kevin Chen, M.D., a neurosurgeon at University of Michigan Health and clinical assistant professor of neurosurgery at U-M Medical School.
“The cells survived for so long with fewer injections and less toxicity from immunosuppression when using monoclonal antibodies. This will enable more experiments and studies of stem cell therapies, bringing more promise for their future in the neurosciences.”
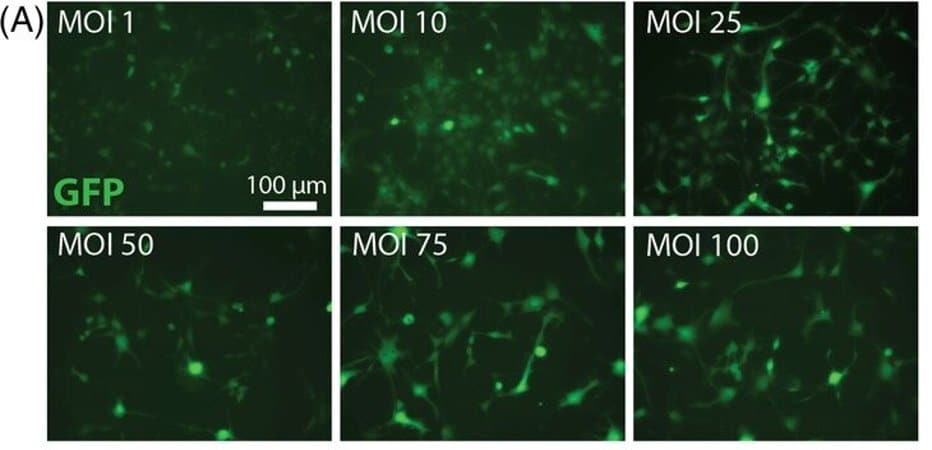
Researchers sought to combat a longstanding obstacle for stem cell therapy in neurological disease of keeping cells alive when testing them in pre-clinical animal models. Many scientists have relied on immunosuppressant medications to keep the animals’ immune systems from rejecting stem cells, Chen says, but they eventually fail and torpedo the process.
“In many of these experiments, we would only see around a third of animals have cells survive and have no way to interpret the results,” he said. “It gets expensive in stem cell therapy to conduct these experiments and not have the cells survive.”
Traditional immunosuppressant drugs are less selective than monoclonal antibodies, which, in this study, targeted two immune proteins. The antibodies have only been analyzed in a handful of stem cell therapy studies for the nervous system. However, this study tracked cell survival for as long as eight months—one of the longest time points published for stem cells in the brain and spinal cord.
“This study lays the groundwork for understanding how transplanted stem cells integrate into the brain,” says senior author Eva Feldman, M.D., Ph.D., James W. Albers Distinguished Professor at U-M, the Russell N. DeJong Professor of Neurology and director of the NeuroNetwork for Emerging Therapies at Michigan Medicine.
“Our new findings continue to support advancing stem cell therapies into human clinical trials,” Feldman said. “Stem cell therapy remains a beacon of hope for neurological diseases.”
About the stem cells research news
Author: Noah Fromson
Source: University of Michigan
Contact: Noah Fromson – University of Michigan
Image: The image is credited to the researchers
Original Research: Open access.
“Monoclonal antibody‐mediated immunosuppression enables long‐term survival of transplanted human neural stem cells in mouse brain” by Lisa M. McGinley et al. Clinical and Translational Medicine
Abstract
Monoclonal antibody‐mediated immunosuppression enables long‐term survival of transplanted human neural stem cells in mouse brain
Background
As the field of stem cell therapy advances, it is important to develop reliable methods to overcome host immune responses in animal models. This ensures survival of transplanted human stem cell grafts and enables predictive efficacy testing. Immunosuppressive drugs derived from clinical protocols are frequently used but are often inconsistent and associated with toxic side effects. Here, using a molecular imaging approach, we show that immunosuppression targeting costimulatory molecules CD4 and CD40L enables robust survival of human xenografts in mouse brain, as compared to conventional tacrolimus and mycophenolate mofetil.
Methods
Human neural stem cells were modified to express green fluorescent protein and firefly luciferase. Cells were implanted in the fimbria fornix of the hippocampus and viability assessed by non-invasive bioluminescent imaging. Cell survival was assessed using traditional pharmacologic immunosuppression as compared to monoclonal antibodies directed against CD4 and CD40L. This paradigm was also implemented in a transgenic Alzheimer’s disease mouse model.
Results
Graft rejection occurs within 7 days in non-immunosuppressed mice and within 14 days in mice on a traditional regimen. The addition of dual monoclonal antibody immunosuppression extends graft survival past 7 weeks (p < .001) on initial studies. We confirm dual monoclonal antibody treatment is superior to either antibody alone (p < .001). Finally, we demonstrate robust xenograft survival at multiple cell doses up to 6 months in both C57BL/6J mice and a transgenic Alzheimer’s disease model (p < .001). The dual monoclonal antibody protocol demonstrated no significant adverse effects, as determined by complete blood counts and toxicity screen.
Conclusions
This study demonstrates an effective immunosuppression protocol for preclinical testing of stem cell therapies. A transition towards antibody-based strategies may be advantageous by enabling stem cell survival in preclinical studies that could inform future clinical trials.






