Summary: Following injury or damage, insulin plays a key role in the maturation and regeneration of immature olfactory sensory neurons.
Source: Monell Chemical Senses Center
Researchers have known for some time that insulin plays a vital role in regeneration and growth in some types of neurons that relay environmental sensory information to our brains, such as sight. However, they know relatively little about the role of insulin in the sense of smell.
Now, investigators at the Monell Chemical Senses Center have shown that insulin plays a critical role in the maturation, after injury, of immature olfactory sensory neurons (OSNs).
The team published their findings in eNeuro earlier this month.
“Our findings suggest that applying insulin into the nasal passage could be developed as a therapy for injury caused by a host of issues,” said first author Akihito Kuboki, MD, a postdoctoral fellow in the lab of Johannes Reisert, PhD.”
Knowing that insulin is part of the body’s repair pathway for visual neurons, Kuboki suspected that the hormone might also play a role in the maturation of OSNs after injury. He also notes there are many insulin receptors in the olfactory region of the brain. Taking these factors into account, Kuboki concluded that insulin may also be involved in the sense of smell.
“Although scientists don’t yet have a clear idea of how it works, we know that insulin plays a key role in preventing cell death,” said Kuboki. “If insulin levels are reduced, diabetes patients have a high susceptibility to cell death, which can cause smell loss.” He is pursuing this research path to shed light on why people with diabetes often suffer from smell loss, or anosmia.
The research team induced diabetes type 1 in mice to reduce levels of circulating insulin reaching the OSNs. The reduced insulin interfered with the regeneration of OSNs, resulting in an impaired sense of smell. They analyzed how the structure of the olfactory tissue in the nasal cavity and the olfactory bulb is impaired by comparing the number of mature OSNs and how well the axons of OSNs reached the olfactory bulb.
The team also recorded odorant-induced responses in the OSNs in the nasal cavity. An odor-guided behavioral task, in which the mice needed to find a cookie reward depending on their ability to smell, measured olfactory function.
In addition, the team injured OSNs, which have a unique ability to regenerate in mammals. This approach allowed the investigators to ask whether OSNs required insulin to regenerate, which they found to be true. What’s more, they discovered that OSNs are highly susceptible to insulin deprivation-induced cell death eight to 13 days after an injury.
This time window indicates that during a critical stage newly generated OSNs are dependent on insulin. They also found that insulin must be applied to regenerating OSNs at this critical time point in the neurons’ growth to be able to restore a mouse’s sense of smell.
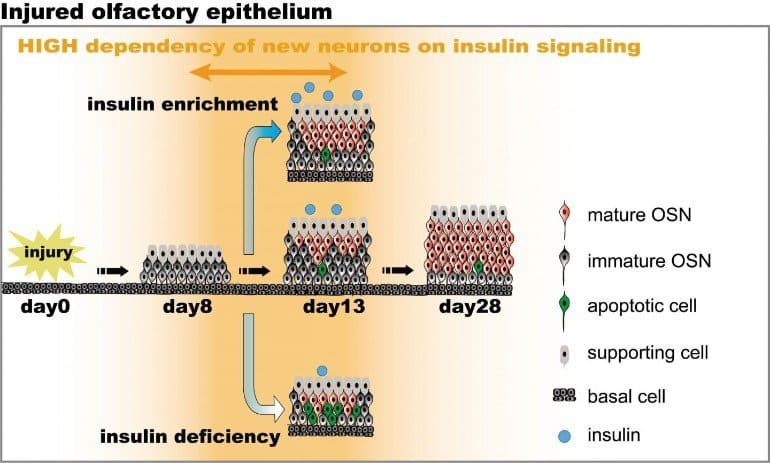
Also of significance, the team found that insulin promotes regeneration of regenerating OSNs in both type 1 diabetic and nondiabetic mice. “Even in nondiabetic mice, we found that insulin can promote the regeneration of OSNs, which suggests that this could be a therapy for olfactory dysfunction in patients without diabetes,” said Kuboki. Specifically, the team only examined the OSN regeneration process after injury in type 1-diabetic mice and did not examine the effects of type 2 diabetes, but plan to in the future.
“Our findings suggest that insulin plays important roles when OSNs need to regenerate after severe injury that induces cell death in many OSNs,” said Kuboki. “From this, we hope that an insulin spray can be potentially applied to treat smell loss for various reasons, including head trauma and viral infection.”
Other members of the research team are Ichiro Matsumoto, PhD, from Monell; Nobuyoshi Otori, MD, PhD and Hiromi Kojima, MD, PhD, from Jikei University School of Medicine; and Shu Kikuta, MD, PhD and Tatsuya Yamasoba, MD, PhD, from the University of Tokyo.
About this neuroscience research news
Source: Monell Chemical Senses Center
Contact: Karen Kreeger – Monell Chemical Senses Center
Image: The image is credited to Monell Chemical Senses Center, eNeuro
Original Research: Closed access.
“Insulin-dependent maturation of newly generated olfactory sensory neurons after injury” by Akihito Kuboki, Shu Kikuta, Nobuyoshi Otori, Hiromi Kojima, Ichiro Matsumoto, Johannes Reisert and Tatsuya Yamasoba. eNeuro
Abstract
Insulin-dependent maturation of newly generated olfactory sensory neurons after injury
Loss of olfactory sensory neurons (OSNs) after injury to the olfactory epithelium (OE) triggers the generation of OSNs that are incorporated into olfactory circuits to restore olfactory sensory perception.
This study addresses how insulin receptor-mediated signaling affects the functional recovery of OSNs after OE injury. Insulin levels were reduced in mice by ablating the pancreatic beta cells via streptozotocin injections. These streptozotocin-induced diabetic and control mice were then intraperitoneally injected with the olfactotoxic drug methimazole to selectively ablate OSNs. The OE of diabetic and control mice regenerated similarly until day 14 after injury. Thereafter, the OE of diabetic mice contained fewer mature and more apoptotic OSNs than control mice.
Functionally, diabetic mice showed reduced electro-olfactogram responses and their olfactory bulbs had fewer c-Fos-active cells following odor stimulation, as well as performed worse in an odor-guided task compared to control mice. Insulin administered intranasally during day 8 to 13 after injury was sufficient to rescue recovery of OSNs in diabetic mice compared to control levels, while insulin administration between days 1 – 6 did not.
During this critical time window on day 8 – 13 after injury, insulin receptors are highly expressed and intranasal application of an insulin receptor antagonist inhibits regeneration. Furthermore, an insulin-enriched environment could facilitate regeneration even in non-diabetic mice.
These results indicate that insulin facilitates the regeneration of OSNs after injury and suggest a critical stage during recovery (8 – 13 days after injury) during which the maturation of newly generated OSNs is highly dependent on and promoted by insulin.
Significance Statement
Although insulin receptor signaling is known to have an influence on cellular processes such as proliferation and apoptosis, it is poorly understood whether the insulin influences the regeneration of olfactory sensory neurons (OSNs) after injury. We compared the maturation processes of new OSNs after the methimazole-induced loss of pre-existing OSNs between diabetic and control mice.
The results show that the regeneration of new OSNs depends on sufficient insulin levels during a specific temporal window, when insulin receptor expression is highly upregulated. Furthermore, an insulin-enriched environment via nasal insulin application during the critical period facilitates OSNs regeneration even in non-diabetic mice.
The present results have implications for intranasal application of insulin as potential clinical therapeutics to facilitate OSNs regeneration after the injury.






