Scientists have uncovered how inflammation and lack of oxygen conspire to cause brain damage in conditions such as stroke and Alzheimer’s disease.
The discovery, published today in Neuron, brings researchers a step closer to finding potential targets to treat neurodegenerative disorders.
Chronic inflammation and hypoxia, or oxygen deficiency, are hallmarks of several brain diseases, but little was known about how they contribute to symptoms such as memory loss.
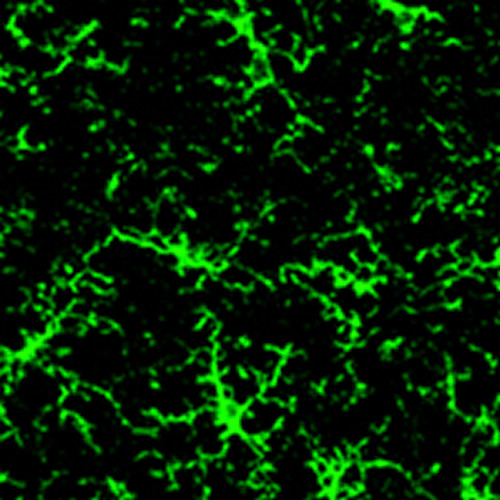
The study used state-of-the-art techniques that reveal the movements of microglia, the brain’s resident immune cells. Brain researcher Brian MacVicar had previously captured how they moved to areas of injury to repair brain damage.
The new study shows that the combination of inflammation and hypoxia activates microglia in a way that persistently weakens the connection between neurons. The phenomenon, known as long-term depression, has been shown to contribute to cognitive impairment in Alzheimer’s disease.
“This is a never-before-seen mechanism among three key players in the brain that interact together in neurodegenerative disorders,” says MacVicar with the Djavad Mowafaghian Centre for Brain Health at UBC and Vancouver Coastal Health Research Institute.
“Now we can use this knowledge to start identifying new potential targets for therapy.”
This video describes the findings, including microscopy footage of how microglia repair brain damage.
Contact: Prof. Brian MacVicar – University of British Columbia
Source: University of British Columbia press release
Image Source: The image is adapted from the University of British Columbia press release.
Video Source: The video “Brain dysfunction mechanism decoded” is available at the ubcpublicaffairs YouTube page.
Original Research: Abstract for “Microglial CR3 Activation Triggers Long-Term Synaptic Depression in the Hippocampus via NADPH Oxidase” by Jingfei Zhang, Aqsa Malik, Hyun B. Choi, Rebecca W.Y. Ko, Lasse Dissing-Olesen, and Brian A. MacVicar in Neuron. Published online March 13 2014 doi:10.1016/j.neuron.2014.01.043






