Summary: Astrocytes harbor HIV and spread the virus to CD4+ T cells that traffic the virus out of the brain and into other organs. HIV moves via this route, even when the virus is suppressed with cART.
Source: NIH/NIMH
Researchers have found that astrocytes, a type of brain cell can harbor HIV and then spread the virus to immune cells that traffic out of the brain and into other organs. HIV moved from the brain via this route even when the virus was suppressed by combination antiretroviral therapy (cART), a standard treatment for HIV. The study, conducted by researchers at Rush University Medical Center in Chicago and published in PLOS Pathogens, was funded by the National Institutes of Health.
“This study demonstrates the critical role of the brain as a reservoir of HIV that is capable of re-infecting the peripheral organs with the virus,” said Jeymohan Joseph, Ph.D., chief of the HIV Neuropathogenesis, Genetics, and Therapeutics Branch at NIH’s National Institute of Mental Health, which co-funded the study. “The findings suggest that in order to eradicate HIV from the body, cure strategies must address the role of the central nervous system.”
HIV attacks the immune system by infecting CD4 positive (CD4+) T cells, a type of white blood cell that is vital to fighting off infection. Without treatment, HIV can destroy CD4+ T cells, reducing the body’s ability to mount an immune response — eventually resulting in AIDS.
cART, which effectively suppresses HIV infections, has helped many people with HIV live longer, healthier lives. But some studies have shown that many patients receiving antiretroviral drugs also show signs of HIV-associated neurocognitive disorders, such as thinking and memory problems. Researchers know that HIV enters the brain within eight days of infection, but less is known about whether HIV-infected brain cells can release virus that can migrate from the brain back into the body to infect other tissues.
The brain contains billions of astrocytes, which perform a variety of tasks — from supporting communication between brain cells to maintaining the blood-brain barrier. To understand whether HIV can move from the brain to peripheral organs, Lena Al-Harthi, Ph.D., and her research team at Rush University Medical Center transplanted HIV-infected or noninfected human astrocytes into the brains of immunodeficient mice.
The researchers found that the transplanted HIV-infected astrocytes were able to spread the virus to CD4+ T cells in the brain. These CD4+ T cells then migrated out of the brain and into the rest of the body, spreading the infection to peripheral organs such as the spleen and lymph nodes. They also found that HIV egress from the brain occurred, albeit at lower levels, when animals were given cART. When cART treatment was interrupted, HIV DNA/RNA became detectable in the spleen — indicating a rebound of the viral infection.
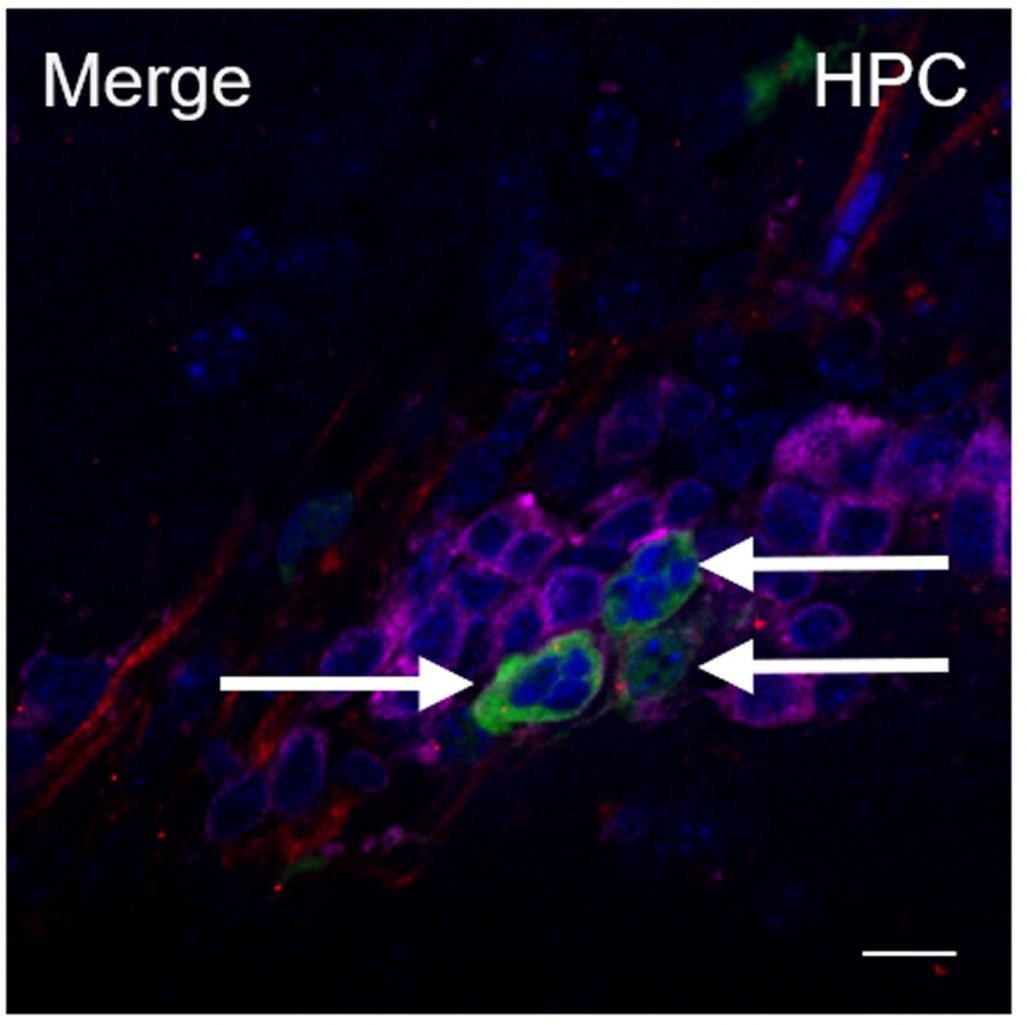
“Our study demonstrates that HIV in the brain is not trapped in the brain — it can and does move back into peripheral organs through leukocyte trafficking,” said Dr. Al-Harthi. “It also shed light on the role of astrocytes in supporting HIV replication in the brain — even under cART therapy.”
This information has significant implications for HIV cure strategies, as such strategies need to be able to effectively target and eliminate reservoirs of HIV replication and reinfection, Dr. Al-Harthi added.
“HIV remains a major global public health concern, affecting 30 to 40 million people across the globe. To help patients, we need to fully understand how HIV affects the brain and other tissue-based reservoirs,” said May Wong, Ph.D., program director for the NeuroAIDS and Infectious Diseases in the Neuroenvironment at the NIH’s National Institute of Neurological Disorders and Stroke, which co-funded the study. “Through additional studies that replicate these findings are needed, this study brings us another step closer towards that understanding.”
About this neuroscience research article
Source:
NIH/NIMH
Media Contacts:
Press Office – NIH/NIMH
Image Source:
The image is credited to Al-Harthi et al. (2020).
Original Research: Open access
“HIV infects astrocytes in vivo and egresses from the brain to the periphery”. by Victoria Lutgen, Srinivas D. Narasipura, Hannah J. Barbian, Maureen Richards, Jennillee Wallace, Roshanak Razmpour, Tetyana Buzhdygan, Servio H. Ramirez, Lisa Prevedel, Eliseo A. Eugenin, Lena Al-Harthi.
PLOS Pathogens doi:10.1371/journal.ppat.1008381
Abstract
HIV infects astrocytes in vivo and egresses from the brain to the periphery
HIV invades the brain during acute infection. Yet, it is unknown whether long-lived infected brain cells release productive virus that can egress from the brain to re-seed peripheral organs. This understanding has significant implication for the brain as a reservoir for HIV and most importantly HIV interplay between the brain and peripheral organs. Given the sheer number of astrocytes in the human brain and their controversial role in HIV infection, we evaluated their infection in vivo and whether HIV infected astrocytes can support HIV egress to peripheral organs. We developed two novel models of chimeric human astrocyte/human peripheral blood mononuclear cells: NOD/scid-IL-2Rgc null (NSG) mice (huAstro/HuPBMCs) whereby we transplanted HIV (non-pseudotyped or VSVg-pseudotyped) infected or uninfected primary human fetal astrocytes (NHAs) or an astrocytoma cell line (U138MG) into the brain of neonate or adult NSG mice and reconstituted the animals with human peripheral blood mononuclear cells (PBMCs). We also transplanted uninfected astrocytes into the brain of NSG mice and reconstituted with infected PBMCs to mimic a biological infection course. As expected, the xenotransplanted astrocytes did not escape/migrate out of the brain and the blood brain barrier (BBB) was intact in this model. We demonstrate that astrocytes support HIV infection in vivo and egress to peripheral organs, at least in part, through trafficking of infected CD4+ T cells out of the brain. Astrocyte-derived HIV egress persists, albeit at low levels, under combination antiretroviral therapy (cART). Egressed HIV evolved with a pattern and rate typical of acute peripheral infection. Lastly, analysis of human cortical or hippocampal brain regions of donors under cART revealed that astrocytes harbor between 0.4–5.2% integrated HIV gag DNA and 2–7% are HIV gag mRNA positive. These studies establish a paradigm shift in the dynamic interaction between the brain and peripheral organs which can inform eradication of HIV reservoirs.
Feel Free To Share This Neuroscience News.






