How does memory occur? And what about movement, or thinking? One key element to understand all these brain functions are the synapses. A synapse is the contact point between two neurons, where a signal is transmitted one-way, from one neuron to another. Specifically, from the pre-synaptic part of one neuron, to the post-synaptic part of another neuron. This communication process involves many types of proteins, and allows us, for example, to memorize a friend’s name. Moreover, synapses do not just pass ‘messages’ from one neuron to another, but also keep the strength of transmitting such ‘messages’ for a short or a long period of time, thus playing a main role in memory formation.
Synaptic malfunction is thought to underlie the early stages of neuro-degenerative diseases, like Parkinson’s and Alzheimer’s diseases. Because of that, understanding the disease progression inside the synapse can lead to the development of drugs to control such diseases. However, a problem when studying synapses is that they are too small for direct access with recording tools. Even with a powerful microscope, it is difficult to see what is happening within a single synapse. But researchers from the Okinawa Institute of Science and Technology Graduate University (OIST) and Doshisha University have found a way around this problem. They have discovered a method to create a synapse that is large enough to allow them to directly monitor events occurring inside the synaptic structure.
In an article published in The Journal of Neuroscience, Dr Dimitar Dimitrov, from the OIST Cellular and Molecular Synaptic Function Unit headed by Prof Tomoyuki Takahashi, and colleagues describe a new method to grow giant synapses in culture dishes. They take fresh neuronal cells from specific regions of mice brains, grow them in a culture dish, and let them form giant synapses. These giant synapses are distinct from conventional small synapses. In these giant synapses, the pre-synaptic part of one neuron does not just ‘touch’ the post-synaptic part of another neuron, but wraps around the whole cell body of the post-synaptic neuron.
This breakthrough was made after many trials and errors. Scientists have studied synaptic mechanisms using fresh brain slices of mice or conventional culture preparations of small synapse. Freshly prepared brain slices can provide researchers with big synapses, but they deteriorate fast, and can be kept and studied only for one day. Such a short period does not allow researchers to perform experiments linked with the molecular composition of neurons and gene expression inside neurons: experiments that involve proteins created inside neurons. To top it all off, brain slices are densely packed with cells and not transparent enough to perform high resolution imaging studies.
Instead, conventional culture preparations are viable for longer periods and their simple cell layer organization makes them ideal for microscopy imaging. But the size of the individual synapse is too small to show in details what is happening inside the synapse.
Dimitrov and colleagues wanted to combine the advantages of the two techniques: big synapses in culture. “Initially, it was not possible to consistently reproduce these big synapses in culture,” Dimitrov recollects. “Then we discovered some specific factors that are needed to promote the formation of the giant synapses.”
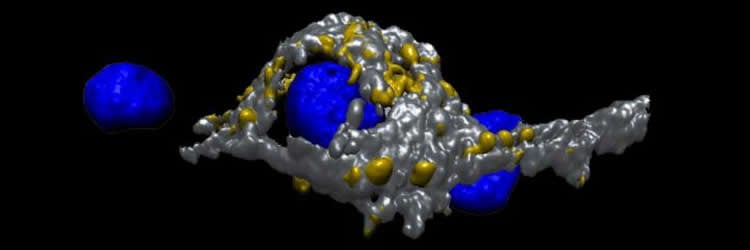
The next step will be to use this model to explore how the proteins relate to the strength of the ‘message’ transmission in the pre-synaptic part of one neuron. To make such studies possible, scientists need to combine microscopic imaging techniques or genetic techniques, which allow them to understand the protein function, with other recording techniques. These latter techniques focus on the electrical proprieties of neurons. In other words, these are electrophysiological techniques that can simultaneously record the transmitted ‘message’.
The giant synapse culture has already shown some encouraging results in this research direction. Scientists were able to record live images of the synapse in high resolution while simultaneously recording the electrical signals that were transmitted between the neurons. “This technique has a huge potential to help us understand how the synapses work. It will provide better, deeper understanding as to what is happening in the synapses,” Prof Takahashi said.
Source: Kaoru Natori – OIST
Image Source: The image is credited to OIST.
Original Research: Abstract for “Reconstitution of Giant Mammalian Synapses in Culture for Molecular Functional and Imaging Studies” by Dimitar Dimitrov, Hiroshi Takagi, Laurent Guillaud, Naoto Saitoh, Kohgaku Eguchi, and Tomoyuki Takahashi in Journal of Neuroscience. Published online March 23 2016 doi:10.1523/JNEUROSCI.3869-15.2016
Abstract
Reconstitution of Giant Mammalian Synapses in Culture for Molecular Functional and Imaging Studies
Giant presynaptic terminal brain slice preparations have allowed intracellular recording of electrical signals and molecular loading, elucidating cellular and molecular mechanisms underlying neurotransmission and modulation. However, molecular genetic manipulation or optical imaging in these preparations is hampered by factors, such as tissue longevity and background fluorescence. To overcome these difficulties, we developed a giant presynaptic terminal culture preparation, which allows genetic manipulation and enables optical measurements of synaptic vesicle dynamics, simultaneously with presynaptic electrical signal recordings. This giant synapse reconstructed from dissociated mouse brainstem neurons resembles the development of native calyceal giant synapses in several respects. Thus, this novel preparation constitutes a powerful tool for studying molecular mechanisms of neurotransmission, neuromodulation, and neuronal development.
SIGNIFICANCE STATEMENT We have developed a novel culture preparation of giant mammalian synapses. These presynaptic terminals make it possible to perform optical imaging simultaneously with presynaptic electrophysiological recording. We demonstrate that this enables one to dissect endocytic and acidification times of synaptic vesicles. In addition, developmental elimination and functional maturation in this cultured preparation provide a useful model for studying presynaptic development. Because this giant synapse preparation allows molecular genetic manipulations, it constitutes a powerful new tool for studying molecular mechanisms of neurotransmission, neuromodulation, and neuronal development.
“Reconstitution of Giant Mammalian Synapses in Culture for Molecular Functional and Imaging Studies” by Dimitar Dimitrov, Hiroshi Takagi, Laurent Guillaud, Naoto Saitoh, Kohgaku Eguchi, and Tomoyuki Takahashi in Journal of Neuroscience. Published online March 23 2016 doi:10.1523/JNEUROSCI.3869-15.2016






