Summary: Two new studies shed light on how the brain encodes and recalls memories.
Source: NIH/NINDS.
Studies of epilepsy patients uncover clues to how the brain remembers.
In a pair of studies, scientists at the National Institutes of Health explored how the human brain stores and retrieves memories. One study suggests that the brain etches each memory into unique firing patterns of individual neurons. Meanwhile, the second study suggests that the brain replays memories faster than they are stored.
The studies were led by Kareem Zaghloul, M.D., Ph.D., a neurosurgeon-researcher at the NIH’s National Institute of Neurological Disorders and Stroke (NINDS). Persons with drug resistant epilepsy in protocols studying surgical resection of their seizure focus at the NIH’s Clinical Center enrolled in this study. To help locate the source of the seizures, Dr. Zaghloul’s team surgically implanted a grid of electrodes into the patients’ brains and monitored electrical activity for several days.
“The primary goal of these recordings is to understand how to stop the seizures. However, it’s also a powerful opportunity to learn how the brain works,” said Dr. Zaghloul.
For both studies, the researchers monitored brain electrical activity while testing the patients’ memories. The patients were shown hundreds of pairs of words, like “pencil and bishop” or “orange and navy,” and later were shown one of the words and asked to remember its pair.
In one study, published in the Journal of Neuroscience, the patients correctly remembered 38 percent of the word pairs they were shown. Electrical recordings showed that the brain waves the patients experienced when they correctly stored and remembered a word pair often occurred in the temporal lobe and prefrontal cortex regions. Nevertheless, the researchers showed that the waves that appeared when recalling the words happened faster than the waves that were present when they initially stored them as memories.
“Our results suggest the brain replays memories on fast forward,” said Dr. Zaghloul.
In the second study, published in Current Biology, the researchers used a new type of grid, called a high density microelectrode array, to monitor the activity of dozens of individual neurons during the memory tests. The arrays were implanted into the middle temporal gyrus, a part of the brain thought to control word, face and distance recognition.
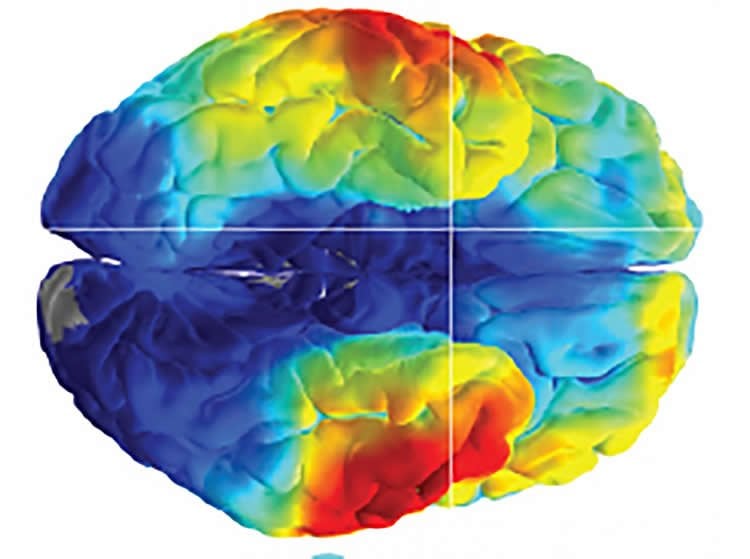
In this study, the patients correctly remembered 23 percent of the word pairs. When the researchers looked at the electrical recordings, they found that the pattern of neurons that fired when the patients correctly recalled a word pair appeared to be similar to the pattern of neurons that fired when they first learned the pair. Moreover, the results showed that the overall activity of the neurons was specific to each individual word pair and was quietest when the patients correctly remembered a pair, suggesting that the brain only uses a small proportion of neurons to represent each memory.
“These results support the idea that each memory is encoded by a unique firing pattern of individual neurons in the brain,” concluded Dr. Zaghloul.
In the future, Dr. Zaghloul’s team plans to continue exploring the neural mechanisms that underlie how the brain forms and retrieves memories and whether they can use similar techniques to understand the electrical codes underlying the epilepsies.
Funding: These studies were supported by NINDS’ Intramural Research Program.
Source: Christopher G. Thomas – NIH/NINDS
Image Source: NeuroscienceNews.com image is credited to Zaghloul lab, NIH/NINDS.
Original Research: Abstract for “Human Cortical Neurons in the Anterior Temporal Lobe Reinstate Spiking Activity during Verbal Memory Retrieval” by Anthony I. Jang, John H. Wittig Jr., Sara K. Inati, and Kareem A. Zaghloul in Current Biology. Published online May 4 2017 doi:10.1016/j.cub.2017.05.014
Abstract for “Cued Memory Retrieval Exhibits Reinstatement of High Gamma Power on a Faster Timescale in the Left Temporal Lobe and Prefrontal Cortex” by Anthony I. Jang, John H. Wittig Jr., Sara K. Inati, and Kareem A. Zaghloul in Current Biology. Published online May 4 2017 doi:10.1016/j.cub.2017.05.014
[cbtabs][cbtab title=”MLA”]NIH/NINDS “Cracking the Brain’s Memory Codes.” NeuroscienceNews. NeuroscienceNews, 2 June 2017.
<https://neurosciencenews.com/epilepsy-memory-codes-6818/>.[/cbtab][cbtab title=”APA”]NIH/NINDS (2017, June 2). Cracking the Brain’s Memory Codes. NeuroscienceNew. Retrieved June 2, 2017 from https://neurosciencenews.com/epilepsy-memory-codes-6818/[/cbtab][cbtab title=”Chicago”]NIH/NINDS “Cracking the Brain’s Memory Codes.” https://neurosciencenews.com/epilepsy-memory-codes-6818/ (accessed June 2, 2017).[/cbtab][/cbtabs]
Abstract
Human Cortical Neurons in the Anterior Temporal Lobe Reinstate Spiking Activity during Verbal Memory Retrieval
Highlights
•Memory retrieval reinstates spiking activity in the human middle temporal gyrus (MTG)
•MTG neurons show spiking responses when forming associations between pairs of words
•Reinstatement of spiking activity is specific when retrieving verbal associations
Summary
When we recall an experience, we rely upon the associations that we formed during the experience, such as those among objects, time, and place. These associations are better remembered when they are familiar and draw upon generalized knowledge, suggesting that we use semantic memory in the service of episodic memory. Moreover, converging evidence suggests that episodic memory retrieval involves the reinstatement of neural activity that was present when we first experienced the event. Therefore, we hypothesized that retrieving associations should also reinstate the neural activity responsible for semantic processing. Indeed, previous studies have suggested that verbal memory retrieval leads to the reinstatement of activity across regions of the brain that include the distributed semantic processing network, but it is unknown whether and how individual neurons in the human cortex participate in the reinstatement of semantic representations. Recent advances using high-density microelectrode arrays (MEAs) have allowed clinicians to record from populations of neurons in the human cortex. Here we used MEAs to record neuronal spiking activity in the human middle temporal gyrus (MTG), a cortical region supporting the semantic representation of words, as participants performed a verbal paired-associates task. We provide novel evidence that population spiking activity in the MTG forms distinct representations of semantic concepts and that these representations are reinstated during the retrieval of those words.
“Human Cortical Neurons in the Anterior Temporal Lobe Reinstate Spiking Activity during Verbal Memory Retrieval” by Robert B. Yaffe, Ammar Shaikhouni, Jennifer Arai, Sara K. Inati and Kareem A. Zaghloul in Journal of Neuroscience. Published online April 26 2017 doi:10.1523/JNEUROSCI.3810-16.2017
Abstract
Cued Memory Retrieval Exhibits Reinstatement of High Gamma Power on a Faster Timescale in the Left Temporal Lobe and Prefrontal Cortex
Converging evidence suggests that reinstatement of neural activity underlies our ability to successfully retrieve memories. However, the temporal dynamics of reinstatement in the human cortex remain poorly understood. One possibility is that neural activity during memory retrieval, like replay of spiking neurons in the hippocampus, occurs at a faster timescale than during encoding. We tested this hypothesis in 34 participants who performed a verbal episodic memory task while we recorded high gamma (62–100 Hz) activity from subdural electrodes implanted for seizure monitoring. We show that reinstatement of distributed patterns of high gamma activity occurs faster than during encoding. Using a time-warping algorithm, we quantify the timescale of the reinstatement and identify brain regions that show significant timescale differences between encoding and retrieval. Our data suggest that temporally compressed reinstatement of cortical activity is a feature of cued memory retrieval.
SIGNIFICANCE STATEMENT We show that cued memory retrieval reinstates neural activity on a faster timescale than was present during encoding. Our data therefore provide a link between reinstatement of neural activity in the cortex and spontaneous replay of cortical and hippocampal spiking activity, which also exhibits temporal compression, and suggest that temporal compression may be a universal feature of memory retrieval.
“Cued Memory Retrieval Exhibits Reinstatement of High Gamma Power on a Faster Timescale in the Left Temporal Lobe and Prefrontal Cortex” by Anthony I. Jang, John H. Wittig Jr., Sara K. Inati, and Kareem A. Zaghloul in Current Biology. Published online May 4 2017 doi:10.1016/j.cub.2017.05.014