Summary: Researchers discover differences in the structure of the cerebral cortex in depressed patients, but the differences can be normalized following taking appropriate medications.
Source: Children’s Hospital Los Angeles.
A study led by Ravi Bansal, PhD, and Bradley S. Peterson, MD, of The Saban Research Institute of Children’s Hospital Los Angeles, has found structural differences in the cerebral cortex of patients with depression and that these differences normalize with appropriate medication. The study, published in Molecular Psychiatry on March 7, is the first to report within the context of a randomized, controlled trial, the presence of structural changes in the cerebral cortex during medication treatment for depression and the first to provide in vivo evidence for the presence of anatomical neuroplasticity in human brain.
“Our findings suggest that thickening of the cerebral cortex is a compensatory, neuroplastic response that helps to reduce the severity of depressive symptoms,” said Peterson, director of the Institute of the Developing Mind at CHLA and professor of pediatrics and psychiatry at the Keck School of Medicine of the University of Southern California. “Patients off medication have a thickened cortex, and the thicker it is, the fewer the symptoms they have. Treatment with medication then reduces the severity of symptoms, which in turn reduces the need for biological compensation in the brain – so that their cortex becomes thinner, reaching thickness values similar to those in healthy volunteers.”
The investigators acquired anatomical brain scans at baseline and again at the end of the 10-week study period for 41 patients with chronic depression, while 39 healthy volunteers were scanned once. This study was conducted with adult patients treated at Columbia University, when Peterson and Bansal were faculty members.
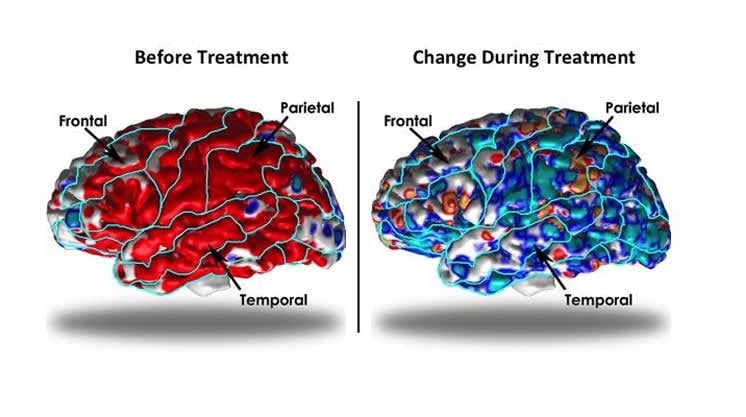
Patients were randomized to receive active medication duloxetine, a selective serotonin and norepinephrine reuptake inhibitor, or placebo. During the trial, patients receiving medication experienced significant improvement of symptoms compared with patients receiving placebo. In medication-treated patients, cortical thickness declined toward values found in healthy volunteers while placebo-treated patients showed a slight thickening of the cortex. According to Bansal, a researcher at CHLA and professor of pediatrics at the Keck School of Medicine of USC, this finding suggests that placebo-treated patients continue to require compensation for their ongoing symptoms.
“Although this study was conducted in adults, the methodology developed – pairing a randomized controlled trial with MRI scanning – can be applied to many other populations in both children and adults,” said Bansal. “Also, our observations of neuroplasticity suggest new biological targets for treatment of persons with neuropsychiatric disorders.”
David J. Hellerstein, MD, of the New York State Psychiatric Institute and Columbia University College of Physicians and Surgeons also contributed to the study.
Funding: Funding was provided in part by NIH grant K02-74677, Eli Lilly, Children’s Hospital Los Angeles and the University of Southern California.
Source: Ellin Kavanagh – Children’s Hospital Los Angeles
Image Source: NeuroscienceNews.com image is credited to Ravi Bansal, PhD.
Original Research: Abstract for “Evidence for neuroplastic compensation in the cerebral cortex of persons with depressive illness” by R Bansal, D J Hellerstein & B S Peterson in Molecular Psychiatry. Published online March 7 2017 doi:10.1038/mp.2017.34
[cbtabs][cbtab title=”MLA”]Children’s Hospital Los Angeles “Brain Architecture Alters to Compensate for Depression.” NeuroscienceNews. NeuroscienceNews, 4 March 2017.
<https://neurosciencenews.com/depression-neurobiology-6209/>.[/cbtab][cbtab title=”APA”]Children’s Hospital Los Angeles (2017, March 4). Brain Architecture Alters to Compensate for Depression. NeuroscienceNew. Retrieved March 4, 2017 from https://neurosciencenews.com/depression-neurobiology-6209/[/cbtab][cbtab title=”Chicago”]Children’s Hospital Los Angeles “Brain Architecture Alters to Compensate for Depression.” https://neurosciencenews.com/depression-neurobiology-6209/ (accessed March 4, 2017).[/cbtab][/cbtabs]
Abstract
Evidence for neuroplastic compensation in the cerebral cortex of persons with depressive illness
We yoked anatomical brain magnetic resonance imaging to a randomized, double-blind, placebo-controlled trial (RCT) of antidepressant medication for 10-week’s duration in patients with dysthymia. The RCT study design mitigated ascertainment bias by randomizing patients to receive either duloxetine or placebo, and it supported true causal inferences about treatment effects on the brain by controlling treatment assignment experimentally. We acquired 121 anatomical scans: at baseline and end point in 41 patients and once in 39 healthy controls. At baseline, patients had diffusely thicker cortices than did healthy participants, and patients who had thicker cortices had proportionately less severe symptoms. During the trial, symptoms improved significantly more in medication—compared with placebo-treated patients; concurrently, thicknesses in medication-treated patients declined toward values in healthy controls, but they increased slightly, away from control values, in placebo-treated patients. Changes in symptom severity during the trial mediated the association of treatment assignment with the change in thickness, suggesting that the beneficial effects of medication on symptom severity were at least partially responsible for normalizing cortical thickness. Together our findings suggest that baseline cortical hypertrophy in medication-free patients likely represented a compensatory, neuroplastic response that attenuated symptom severity. Medication then reduced symptoms and lessened the need for compensation, thereby normalizing thickness. This is to the best of our knowledge the first study to report within an RCT a differential change in cortical morphology during medication treatment for depressive illness and the first to provide within an RCT in vivo evidence for the presence of neuroanatomical plasticity in humans.
“Evidence for neuroplastic compensation in the cerebral cortex of persons with depressive illness” by R Bansal, D J Hellerstein & B S Peterson in Molecular Psychiatry. Published online March 7 2017 doi:10.1038/mp.2017.34






