Summary: A multiyear study reveals, contrary to expectations, numbers and ratios of three major inhibitory cell types vary stereotypically across different cortical parts. Additionally, researchers identified 11 subcortical areas with gender specific differences in mice.
Source: Cold Spring Harbor Laboratory.
Neuroscientists at Cold Spring Harbor Laboratory (CSHL) have mobilized advanced imaging and computational methods to comprehensively map – “count” – the total populations of specific types of cells throughout the mouse brain. In a study published today in Cell, they report two highly surprising findings.
Their “qBrain” (quantitative brain) study reveals that contrary to expectations, the numbers and ratios of three major inhibitory cell types vary in a stereotypical way across different parts of the mouse cortex. This implies that different cortical areas, for instance those involving cognition vs. those involving perception of sensory stimuli, have evolved to tailor their local circuits to specific brain functions. Also surprising was that while male and female brains did not differ in cell counts in cortical regions, the study identified 11 subcortical areas with gender-specific differences. Strikingly, despite the overall tendency for male brains to be larger, 10 of these regions had more modulatory neurons in females than in males.
“This shows that there are more cells that modulate signals and exert temporal control in areas regulating reproductive, social and parenting behaviors in females than in males, with one exception so far,” says CSHL Associate Professor Pavel Osten, the team leader. The exception is a small area in the hypothalamus, called posterodorsal preoptic nucleus, that is believed to control a single male-specific reproductive function (ejaculation).
The inhibitory cell types (which modulate the signals of excitatory neurons) counted by the qBrain technology included cells expressing parvalbumin (PV+), somatostatin (SST+) and vasoactive intestinal peptide (VIP+) and four subtypes. Each is a protein that serves to flag a distinct cell type in the mammalian brain. It’s the first step in a project that CSHL and collaborators are performing as part of the National Institute of Health’s “BRAIN Initiative Cell Census Network (BICCN).” In the next 5 years, the team will be building a comprehensive online resource database for the scientific community that will include distribution maps and cellular morphologies for over 100 distinct cell types throughout the mouse brain.
“Even our first results show that it really matters to know the composition of the brain in a precise, quantitative way,” says Osten. qBrain is built on an automated technology platform that will be used to perform similar analyses of other mammalian brains, from prairie voles to marmoset monkeys and humans. This will enable unprecedented cross-species comparisons.
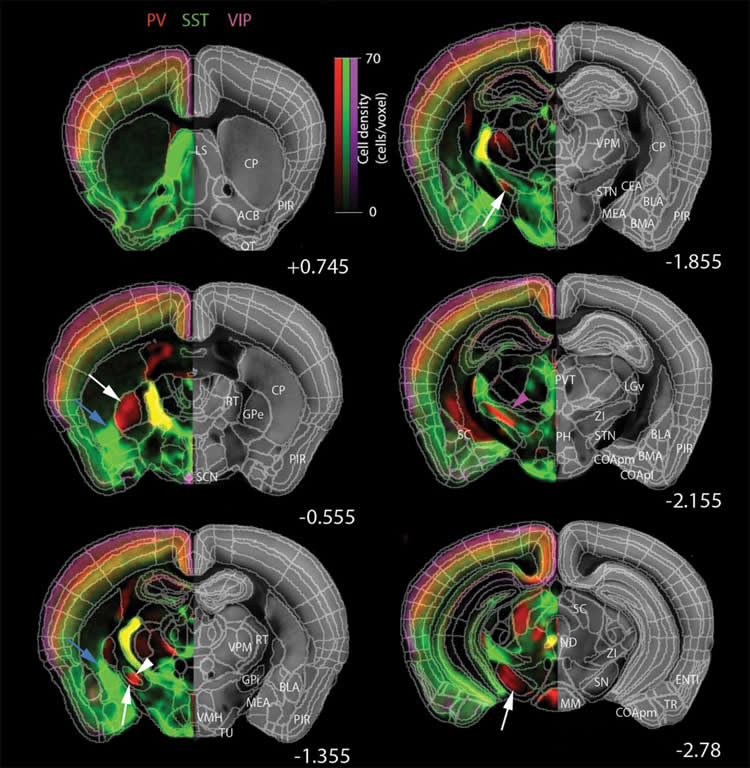
“The brain is like a very complicated Lego puzzle, with pieces that come in all sorts of shapes and sizes,” Osten says. “If you want to understand how brain circuits work, you first need to know how many pieces there are, of what types, and how are they distributed. It’s still not known, but there are thought to be at least 500 cell types in the mammalian brain, and possibly thousands. We still lack a basic parts list.”
CSHL Professors Z. Josh Huang and Partha Mitra collaborated on the qBrain paper. Each mouse brain was scanned and precisely registered in a 600-region grid to a master file by computers, one brain per day. The ~200 GB of data for each brain was then analyzed with machine learning algorithms that identify individual neurons by type, according to parameters “learned” from human experts.
Osten hopes that using qBrain to scan and quantify the brain in mouse models of psychiatric, neurodevelopmental and neurodegenerative disorders – from depression and schizophrenia to autism and Alzheimer’s disease – will provide instructive comparisons with the data drawn from brains of healthy mice. This might enable scientists to begin to clarify what brain irregularities characterize those illnesses.
Funding: This research was supported by the NIH, the Simons Foundation Autism Research Initiative, STCSM, the Pennsylvania Department of Health Tobacco CURE Funds, Samuel J. and Joan B. Williamson Fellowship, Mathers Charitable Foundation, H N Mahabala Chair Professorship and, and IIT Madras.
Source: Peter Tarr – Cold Spring Harbor Laboratory
Image Source: NeuroscienceNews.com image is credited to Osten Lab, CSHL.
Original Research: Full open access research for “Brain-wide Maps Reveal Stereotyped Cell-Type-Based Cortical Architecture and Subcortical Sexual Dimorphism” by Yongsoo Kim8, Guangyu Robert Yang8, Kith Pradhan, Kannan Umadevi Venkataraju, Mihail Bota, Luis Carlos García del Molino, Greg Fitzgerald, Keerthi Ram, Miao He, Jesse Maurica Levine, Partha Mitra, Z. Josh Huang, Xiao-Jing Wang, and Pavel Osten in Cell. Published online October 5 2017 doi:10.1016/j.cell.2017.09.020
[cbtabs][cbtab title=”MLA”]Cold Spring Harbor Laboratory “Mouse Cell Type Census Reveals Gender Specific Brain Differences.” NeuroscienceNews. NeuroscienceNews, 5 October 2017.
<https://neurosciencenews.com/cell-census-brain-structure-7670/>.[/cbtab][cbtab title=”APA”]Cold Spring Harbor Laboratory (2017, October 5). Mouse Cell Type Census Reveals Gender Specific Brain Differences. NeuroscienceNews. Retrieved October 5, 2017 from https://neurosciencenews.com/cell-census-brain-structure-7670/[/cbtab][cbtab title=”Chicago”]Cold Spring Harbor Laboratory “Mouse Cell Type Census Reveals Gender Specific Brain Differences.” https://neurosciencenews.com/cell-census-brain-structure-7670/ (accessed October 5, 2017).[/cbtab][/cbtabs]
Abstract
Brain-wide Maps Reveal Stereotyped Cell-Type-Based Cortical Architecture and Subcortical Sexual Dimorphism
Highlights
•Quantitative maps reveal cell-type-based structural organization of the brain
•PV+ and SST+ interneuron distribution defines cortical hierarchies
•Circuit architectures differ in frontal/ associational and sensorimotor cortices
•SST+ and VIP+ distribution identifies sexually dimorphic subcortical circuits
Summary
The stereotyped features of neuronal circuits are those most likely to explain the remarkable capacity of the brain to process information and govern behaviors, yet it has not been possible to comprehensively quantify neuronal distributions across animals or genders due to the size and complexity of the mammalian brain. Here we apply our quantitative brain-wide (qBrain) mapping platform to document the stereotyped distributions of mainly inhibitory cell types. We discover an unexpected cortical organizing principle: sensory-motor areas are dominated by output-modulating parvalbumin-positive interneurons, whereas association, including frontal, areas are dominated by input-modulating somatostatin-positive interneurons. Furthermore, we identify local cell type distributions with more cells in the female brain in 10 out of 11 sexually dimorphic subcortical areas, in contrast to the overall larger brains in males. The qBrain resource can be further mined to link stereotyped aspects of neuronal distributions to known and unknown functions of diverse brain regions.
“Brain-wide Maps Reveal Stereotyped Cell-Type-Based Cortical Architecture and Subcortical Sexual Dimorphism” by Yongsoo Kim8, Guangyu Robert Yang8, Kith Pradhan, Kannan Umadevi Venkataraju, Mihail Bota, Luis Carlos García del Molino, Greg Fitzgerald, Keerthi Ram, Miao He, Jesse Maurica Levine, Partha Mitra, Z. Josh Huang, Xiao-Jing Wang, and Pavel Osten in Cell. Published online October 5 2017 doi:10.1016/j.cell.2017.09.020






