Summary: Study reveals the significant role lipids play in regulating the amyloid pathway protein C99 and the progression of Alzheimer’s disease.
Source: American Institute of Physics
Alzheimer’s disease is predominant in elderly people, but the way age-related changes to lipid composition affect the regulation of biological processes is still not well understood. Links between lipid imbalance and disease have been established, in which lipid changes increase the formation of amyloid plaques, a hallmark of Alzheimer’s disease.
This imbalance inspired researchers from Aarhus University in Denmark to explore the role of lipids comprising the cellular membranes of brain cells.
In Biointerphases, by AIP Publishing, the researchers report on the significant role lipids may play in regulating C99, a protein within the amyloid pathway, and disease progression. Lipids have been mostly overlooked from a therapeutic standpoint, likely because their influence in biological function is not yet fully understood.
Toxic amyloid plaques are formed within the brain when a series of enzymes cleave the protein APP, which sits within the neuronal cell membrane, to form C99, which in turn is cleaved to release the amyloid-beta peptide that can form plaques.
Both C99 and APP are able to protect themselves from cleavage by forming homodimers, a protein composed of two polypeptide chains that are identical. The interaction between C99 molecules is regulated by lipids that make up the membrane in which the protein sits.
“We showed that a change in the cholesterol content of the neuronal cell membrane can change how the C99 dimerizes,” said Amanda Dyrholm Stange, one of the authors. “Our work suggests age-related changes to cholesterol content in the membrane weakens the C99-C99 interaction, which consequently decreases the ‘protective’ effect of the dimerization process, leading to the hypothesis of why more toxic amyloid-beta peptides are released in the elderly.”
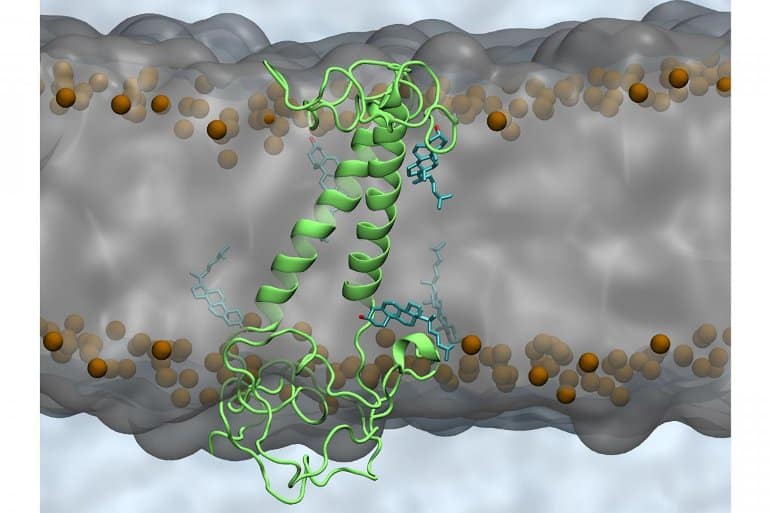
Therapeutics for Alzheimer’s disease currently “have a very high failure rate, with no therapeutics developed for a very long period of time, so a novel strategy is desperately needed,” said co-author Nils Anton Berlund. “Attempting to modulate the composition of the lipid membrane would be an entirely new class of Alzheimer’s disease therapeutics but also immensely challenging without side effects.”
The researchers postulate shifting the strategy away from targeting proteins to instead targeting the lipid concentration of membranes may be worthwhile.
“We hope our work will lead the pharmaceutical/biotechnology sector to choose lipid modulation as a means for targeting in drug development, because these changes in lipid composition are linked not just to Alzheimer’s but a large host of diseases — from diabetes to cardiovascular disease,” said co-author Birgit Schiøtt. “We also hope it will lead to more research and funding toward understanding the fundamental science behind the possible regulatory roles of lipids.”
About this Alzheimer’s disease research news
Source: American Institute of Physics
Contact: Larry Frum – American Institute of Physics
Image: The image is credited to Amanda Dyrholm Stange, Jenny Pin-Chia Hsu, Lisbeth Ravnkilde, Nils Berglund, and Birgit Schiøtt
Original Research: Open access.
“The effect of cholesterol on the dimerization of C99–a molecular modeling perspective” by Amanda Dyrholm Stange, Jenny Pin-Chia Hsu, Lisbeth Ravnkilde Kjølbye, Nils Anton Berglund, and Birgit Schiøtt. Biointerphases
Abstract
The effect of cholesterol on the dimerization of C99–a molecular modeling perspective
C99, a naturally occurring peptide, is a precursor of the amyloid β-peptide (Aβ) and plays an important role in the so-called amyloidogenic pathway of degradation of amyloid precursor protein.
While the effect of C99’s dimerization is not clearly determined, it has been hypothesized that the dimerization protects C99 from being cleaved further. Cholesterol (CHOL) is known to interact with C99 and its presence in high concentrations has been linked to an increase in the production of Aβ; however, to what extent this is correlated, and how, has not yet been determined. In this study, we systematically examine the effect of increasing cholesterol concentration on the homodimerization propensity of C99, combining unbiased atomistic molecular dynamics simulations with biased simulations using a coarse grained resolution.
Through the use of umbrella sampling, we show how the presence of high levels of CHOL destabilizes the interaction between two C99 monomers. The interaction pattern between the two C99s has shifted several residues, from the N-terminal end of the transmembrane region toward the corresponding C-terminal in the presence of CHOL.
The umbrella sampling shows that the presence of high levels of CHOL led to a decrease of the disassociation energy by approximately 3 kJ/mol. In conclusion, this suggests that increasing CHOL destabilizes the interaction between the two C99 monomers, which may possibly cause an increase in the production of Aβ42.






