Summary: A new imaging system has allowed researchers to map whole brain distribution of senile plaques in mouse models of Alzheimer’s disease. The technique could help propel Alzheimer’s research and speed up the development of new treatments.
Source: The Optical Society.
Micron-level, 3-D visualization of protein deposits found in Alzheimer’s disease could speed development of new treatments.
An estimated 5.5 million Americans live with Alzheimer’s disease, a type of dementia that causes problems with memory, thinking and behavior. Although treatments can slow the worsening of symptoms, scientists are still working to better understand the neurodegenerative disease so that curative and preventative medicines can be developed. A new imaging system could help speed new drug development by offering a better way to monitor the brain changes indicative of Alzheimer’s in mouse models of the disease.
Mice genetically modified to exhibit characteristics of Alzheimer’s disease are a valuable tool for studying the disease’s biology and testing new drugs. Like people with the disease, the brains of these mice accumulate clumps of proteins known as senile plaques. Qingming Luo’s Visible Brain-wide Networks team at the Huazhong University of Science and Technology, China developed a system called cryo-micro-optical sectioning tomography (cryo-MOST) that improves the ability to image these senile plaques in the whole mouse brain.
“Studying the brain-wide distribution of senile plaques in mice will facilitate an understanding of how brain functions deteriorate during Alzheimer’s disease progression,” said Jing Yuan, a key member of the research team. “We hope that cryo-MOST will accelerate the development and evaluation of Alzheimer’s disease treatments.”
In The Optical Society (OSA) journal Optics Letters, the researchers detail their cryo-MOST system and report results from using it to create 3D, micron-resolution maps of senile plaques throughout the brain of a mouse model of Alzheimer’s disease. The new system is simple and efficient compared with conventional approaches, requires no external dyes or labels, and because it is optical, offers more detailed information than other imaging techniques such as magnetic resonance imaging (MRI) or Positron Emission Tomography (PET).
Chilling tissue to enhance imaging
Labels are not needed with cryo-MOST because it takes advantage of the natural fluorescence of senile plaques after exposure to excitation light, a property known as autofluorescence. The researchers discovered that lowering the tissue temperature to less than -100 degrees Celsius brightened the autofluorescence from senile plaques and improved the resulting images.
“Exogenous dyes may lead to unspecific, false-positive or uneven labeling in brain tissue, which impedes observation of authentic pathological structure changes of Alzheimer’s disease,” said Yuan. “Our label-free approach avoids these problems while also simplifying the sample preparation, thus accelerating the research process.”
To maintain the ultra-low temperatures required to enhance the fluorescence, the researchers created a system that allowed imaging of a sample immersed in liquid nitrogen. Because traditional optical microscopy can only image the surface of the tissue, they incorporated a mechanical milling machine that removed a layer of the tissue each time the surface was imaged. When acquiring images of the whole mouse brain, the system automatically alternates between milling and imaging by using a mechanical stage to move the sample.
Mapping the senile plaques
To demonstrate the ability of cryo-MOST to image the brain-wide distribution of senile plaques, the researchers used it to image a whole brain from a 17-month APP/PS1 mouse model of Alzheimer’s disease.
“The images from an aged Alzheimer’s disease mouse revealed that senile plaques have spread to the whole brain,” said Yuan. “This indicates that the disease not only hurts memory and intelligence, but may also cause an overall deterioration of other brain functions.”
The system used in the paper has a lateral resolution of 1.072 microns and an axial resolution of 17.152 microns when detecting fluorescence at a wavelength of 536 nm. The researchers say that these parameters could be further improved by using a better microscope.
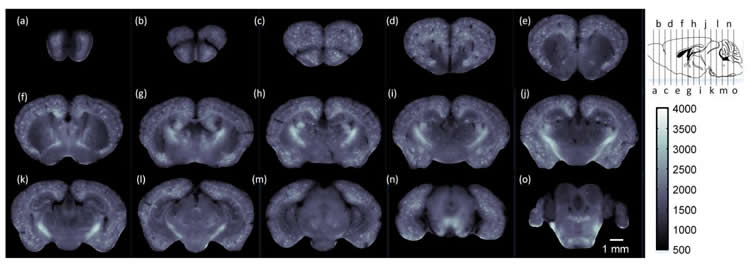
Because the size of the tissue imaged is only limited by the maximum moving range of the mechanical stage, the system could be used for studying human brain tissue from deceased donors. It could also be useful for visualizing other biological molecules that exhibit autofluorescence in other organs. For example, it could image metabolism distribution in organs such as the kidney and liver.
The research team is currently working to improve the automation of the approach to speed up image acquisition and is incorporating enhancements that will improve image quality. These upgrades will allow their approach to be used for more applications. They are also continuing to use cryo-MOST to study senile plaque distribution and morphology changes that come with the progression of Alzheimer’s disease.
“In addition to evaluating drug efficacy for neurodegenerative diseases, we believe our method will help neuroscientists understand the relationship between senile plaque distribution and other key factors of Alzheimer’s disease,” said Yuan. “This will enhance the comprehensive understanding of the disease mechanisms and treatment.”
Source: Joshua Miller – The Optical Society
Publisher: Content organized by NeuroscienceNews.com.
Image Source: NeuroscienceNews.com images is credited to Jing Yuan, Huazhong University of Science and Technology.
Original Research: Abstract for “Label-free brainwide visualization of senile plaque using cryo-micro-optical sectioning tomography” by Yilin Luo, Anle Wang, Mengmeng Liu, Tian Lei, Xiaochuan Zhang, Zhaobing Gao, Hualiang Jiang, Hui Gong, and Jing Yuan in Optics Letters. Published online October 2017 doi:10.1364/OL.42.004247
[cbtabs][cbtab title=”MLA”]The Optical Society “New Imaging Approach Maps Whole Brain Changes From Alzheimer’s: Mouse Study.” NeuroscienceNews. NeuroscienceNews, 17 October 2017.
<https://neurosciencenews.com/brain-mapping-alzheimers-7749/>.[/cbtab][cbtab title=”APA”]The Optical Society (2017, October 17). New Imaging Approach Maps Whole Brain Changes From Alzheimer’s: Mouse Study. NeuroscienceNews. Retrieved October 17, 2017 from https://neurosciencenews.com/brain-mapping-alzheimers-7749/[/cbtab][cbtab title=”Chicago”]The Optical Society “New Imaging Approach Maps Whole Brain Changes From Alzheimer’s: Mouse Study.” https://neurosciencenews.com/brain-mapping-alzheimers-7749/ (accessed October 17, 2017).[/cbtab][/cbtabs]
Abstract
Label-free brainwide visualization of senile plaque using cryo-micro-optical sectioning tomography
Optical visualization of pathological changes in Alzheimer’s disease (AD) can facilitate exploration of disease mechanisms and treatments. However, existing optical imaging methods have limitations on mapping pathological evolution in the whole mouse brain. Previous research indicated endogenous fluorescence contrast of senile plaques. Therefore, we develop cryo-micro-optical sectioning tomography (cryo-MOST) to capture intrinsic fluorescence distribution of senile plaques at a micrometer-level resolution in the whole brain. Validation using immunofluorescence demonstrates the capacity of cryo-MOST to visualize and distinguish senile plaques with competent sensitivity and spatial resolution. Compared with imaging in room temperature, cryo-MOST provides better signal intensity and signal-to-noise ratio. Using cryo-MOST, we obtained whole-brain coronal distribution of senile plaques in a transgenic mouse without exogenous dye. Capable of label-free brainwide visualization of Alzheimer’s pathology, cryo-MOST may be potentially useful for understanding neurodegenerative disease mechanisms and evaluating drug efficacy.
“Label-free brainwide visualization of senile plaque using cryo-micro-optical sectioning tomography” by Yilin Luo, Anle Wang, Mengmeng Liu, Tian Lei, Xiaochuan Zhang, Zhaobing Gao, Hualiang Jiang, Hui Gong, and Jing Yuan in Optics Letters. Published online October 2017 doi:10.1364/OL.42.004247