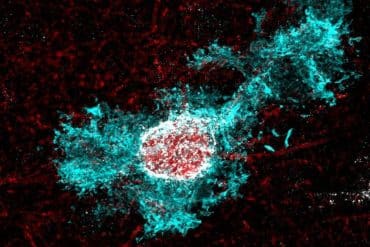
Summary: For over a century, neurons have been considered the sole architects of long-distance communication in the brain. However, a study reveals that star-shaped support cells called astrocytes build their own intricate, organized, and far-reaching networks.
Using a custom-built tracing tool, researchers mapped active astrocyte “webs” that connect distant brain regions, sometimes linking areas that aren’t even joined by neurons. This discovery adds an entirely new layer to our understanding of how the brain stays connected, develops, and responds to disease.
Key Facts
- Beyond Support: Astrocytes, long viewed as mere “utility” cells that ferry nutrients and waste, are now proven to run their own highly specific, brain-wide signaling pathways.
- Gap Junction Bridges: These networks rely on physical channels called gap junctions. When these junctions were genetically removed in mice, the communication webs vanished, proving the pathways are active and structural.
- Experience-Driven Plasticity: The networks are dynamic and “learn” from the environment. When researchers altered sensory input (trimming whiskers), the astrocyte pathways physically rerouted and reconnected to new partners.
- Implications for Disease: Because these networks redistribute resources to damaged areas, they may offer fresh insights into neurodegenerative conditions like Alzheimer’s, Parkinson’s, and glaucoma.
Source: NYU Langone
Cells long thought to play a secondary role in brain function build their own far-reaching connections, a new study in mice showed. These pathways appear to connect distant regions in ways that had not been mapped before.
Experts usually describe the brain as a network of nerve cells (neurons) that send each other signals to pass along information. These neurons are maintained by another kind of brain cell, the star-shaped astrocyte, which ferries in nutrients and carries away waste.
Led by NYU Langone Health researchers, the study revealed that, like neurons, astrocytes form organized webs, which enables them to communicate with other specific astrocytes across the brain rather than only sending local, generalized signals. In some cases, the pathways link areas that were not already joined together by neurons.
“For more than a century, neuroscientists have thought of neurons as the main actors in the brain,” said study lead author Melissa Cooper, PhD. “Yet our findings suggest that astrocytes, which are usually viewed as merely support cells, are also running their own widespread signaling pathway, adding another layer to how brain regions stay connected.”
In earlier work, Dr. Cooper reported that in a mouse model of the visual neurodegenerative disease glaucoma, astrocytes can redistribute resources from astrocytes around healthy neurons to damaged neurons. Yet the team had no way to see whether this kind of support-cell network extended across the entire brain.
This latest investigation is the first to map active, brain-wide communication networks built by astrocytes and to show that these pathways are highly specific, said Dr. Cooper, a postdoctoral fellow in the Department of Neuroscience at NYU Grossman School of Medicine.
The findings, which will publish April 22 in the journal Nature, relied on a custom-built tracing tool that let the team follow the cells’ connections in far greater detail than past methods allowed.
For the study, the researchers used a harmless virus to deliver “network tracers” into astrocytes in selected brain regions of lab mice. These tracers tagged small molecules as the molecules passed through tiny channels called gap junctions, which link one astrocyte to another, allowing the team to see which cells were part of the same signaling pathway.
The scientists then made the mice’s brains transparent and used a specialized microscope to capture three-dimensional images of every tagged astrocyte. By doing this across hundreds of mice, they could map astrocyte webs across brain areas. The tracing tool and brain-clearing method were designed to be relatively low-cost and easy to reproduce so other labs could use them to study the networks in many brain diseases.
In another part of the study, the team assessed mice that were genetically engineered to have astrocytes that lacked gap junctions. The communication networks largely disappeared, suggesting that the pathways are active and depend on these physical bridges.
“By challenging our understanding of how the brain communicates over long distances, our results may offer fresh insight into how it develops, ages, and behaves in conditions such as Alzheimer’s and Parkinson’s disease,” said study co-senior author Shane A. Liddelow, PhD. Dr. Liddelow is an associate professor in the neuroscience and ophthalmology departments at NYU Grossman School of Medicine.
Another key finding was that astrocyte networks are dynamic. When the team trimmed whiskers on one side of the mice’s faces, a pathway from the region that processes whisker touch got smaller and reconnected to different astrocyte partners.
“The fact that astrocyte networks shrink and reroute after a loss of sensory signals suggests they may be shaped by experience,” said study co-senior author Moses V. Chao, PhD.
“It also raises the possibility that each of us has a somewhat unique pattern of connections molded by what our brains have learned and lived through,” added Dr. Chao, a professor in the cell biology, neuroscience, and psychiatry departments at NYU Grossman School of Medicine.
The authors plan to investigate which molecules move through the networks and to apply their tracing tool to models of brain disorders. They also hope to examine how these webs change during development and aging, said Dr. Chao.
Dr. Liddelow emphasized that while gap junctions and astrocytes exist in humans, it remains unknown whether the networks link the same regions in the same way as in mice.
Funding: Funding for the study was provided by National Institutes of Health grants R01EY033353, U19NS107616, P30AG066512, P30CA016087, T32MH019524, K99NS139313, and K00AG068343.
Further funding was provided by Cure Alzheimer’s Fund, the Leon Levy Scholarships in Neuroscience at The New York Academy of Sciences, the Pew Charitable Trusts postdoctoral fellowship, the Simons Foundation SURFiN fellowship, the Belfer Neurodegeneration Consortium, the Carol and Gene Ludwig Family Foundation, and the Swiss National Science Foundation.
Dr. Liddelow maintains a financial interest in AstronauTx Ltd., a company investigating possible treatment targets for Alzheimer’s disease, and Synapticure, a telehealth company that provides care to patients with Alzheimer’s disease, dementia, and other neurological conditions.
He is also on the scientific advisory board of the Global BioAccess Fund. None of these activities is related to the current study. The terms and conditions of these relationships are being managed by NYU Langone Health in accordance with its policies and procedures.
Along with Drs. Cooper, Liddelow, and Chao, other NYU Langone researchers involved in the study are Maria Clara Selles, PhD; Michael Cammer, MFA, MAT; Holly Gildea, PhD; Joseph Sall; and Katelyn Chiurri. Other study co-investigators are Chase Redd, Philip Cheung, MD, and Damian Wheeler, PhD, at Translucence Biosystems in Irvine, California; and Aiman Saab, PhD, at the University of Zurich in Switzerland.
Key Questions Answered:
A: While neurons use electrical impulses and synapses, astrocytes use tiny channels called gap junctions to pass small molecules directly from one cell to another. This creates a “bucket brigade” of information that can travel across large distances of the brain.
A: Yes. Because the study showed that astrocyte networks shrink and reroute based on sensory experience, it suggests that your life history, what you’ve learned, and what you’ve lived through may have molded a unique pattern of astrocyte connections in your brain.
A: Technology was the barrier. Astrocytes are densely packed and their signals are subtle compared to the “loud” electrical spikes of neurons. The NYU team had to develop a specific virus-based tracer and a “brain-clearing” method to make the tissue transparent just to see these webs.
Editorial Notes:
- This article was edited by a Neuroscience News editor.
- Journal paper reviewed in full.
- Additional context added by our staff.
About this AI and auditory neuroscience research news
Author: Shira Polan
Source: NYU Langone Health
Contact: Shira Polan – NYU Langone Health
Image: The image is credited to Neuroscience News
Original Research: Open access.
“Astrocytes connect specific brain regions through plastic networks” by Melissa L. Cooper, Maria Clara Selles, Michael Cammer, Chase Redd, Holly K. Gildea, Joseph Sall, Katelyn E. Chiurri, Philip Cheung, Damian G. Wheeler, Aiman S. Saab, Shane A. Liddelow & Moses V. Chao. Nature
DOI:10.1038/s41586-026-10426
Abstract
Astrocytes connect specific brain regions through plastic networks
Neuronal axons have traditionally been considered to be the primary mediators of functional connectivity among brain regions. However, the role of astrocyte-mediated communication has been largely underappreciated.
Astrocytes communicate with one another through gap junctions, but the extent and specificity of this communication remain poorly understood. Astrocyte gap junctions are necessary for memory formation, synaptic plasticity, coordination of neuronal signalling, and closing the visual and motor critical periods.
These findings indicate that this form of communication is essential for proper central nervous system development and function. Despite the importance of astrocyte gap junctional networks, studying them has been challenging. Current methods such as slice electrophysiology disrupt network connectivity and introduce artefacts due to tissue damage.
Here, we developed a vector-based approach that labels molecules as they are fluxed by astrocyte gap junctions in awake, behaving animals to overcome these limitations.
We then used whole-brain tissue clearing to image these intact, three-dimensional astrocyte networks. We show that multiple astrocyte networks traverse the mouse brain. These networks selectively connect specific regions, rather than diffusing indiscriminately, and vary in size and organization.
We observe local networks that are confined to single brain regions and long-range networks that robustly interconnect multiple regions across hemispheres, often exhibiting patterns distinct from known neuronal networks. We also demonstrate that astrocyte networks undergo structural reorganization in the adult brain after sensory deprivation.
These findings reveal a mode of communication between distant brain regions that is mediated by plastic networks of gap junction-coupled astrocytes.