Summary: The FXR1 protein in the brains of healthy, young rats functions in an amyloid form. The protein has previously been linked to controlling long-term memory formation and emotion.
Source: St. Petersburg State University
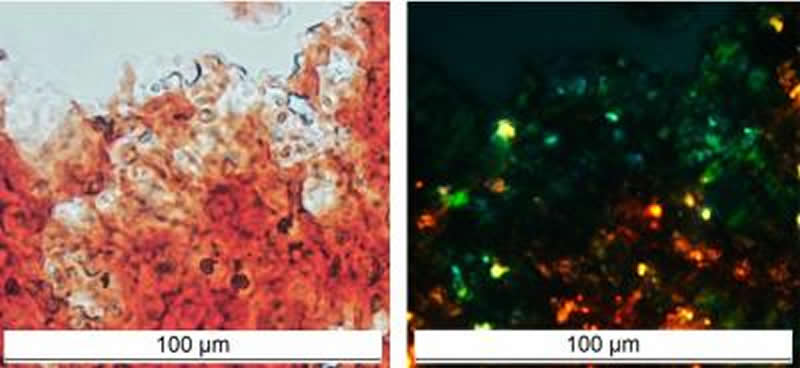
Scientists from St Petersburg University worked with their colleagues from the St Petersburg branch of the Vavilov Institute of General Genetics. They conducted experiments on laboratory rats and showed that the FRX1 protein in the brains of young and healthy animals functions in an amyloid form. The previously published reports indicate that this protein controls long term memory and emotions: mice that have the FRX1 gene “off” quickly remember even complex mazes, and animals that have too much of this protein do not suffer from depression even after severe stress. In addition, in humans, a failure in the gene encoding FRX1 is linked to autism and schizophrenia.
“Our findings clearly show that developing a universal remedy that will destroy all amyloids in the brain is totally futile. Instead, we need to look for a cure for each specific pathology. The healthy brain was previously known to store only a few protein hormones in amyloid form. They are stored in secretory granules in the hypophysis, but when the time comes, the secretory granules burst and the proteins function in a normal, monomeric form,” said Alexey Galkin, Professor of the Department of Genetics, Doctor of Biology. “We have initially proved that the protein can actually function in the brain in amyloid form, both as oligomers and as insoluble aggregates. Also, the amyloid form FRX1 can bind RNA molecules and protect them from degradation.’
The research was conducted by the Research Park of St Petersburg University with equipment provided by the resource centres “Chromas Core Facility” and “The Centre for Molecular and Cell Technologies”. The amyloid form of FXR1 protein was discovered by scientists using the amyloid proteome screening method developed by a research team in 2016. Amyloids generally play an important role in many organisms: for example, one of these proteins is found in human pigment cells and affects skin tanning. However, today, scientists are interested in amyloids primarily due to the need to find a cure for neurodegenerative diseases, where these proteins play a key role.
Source:
St. Petersburg State University
Media Contacts:
Polina Ogorodnikova – St. Petersburg State University
Image Source:
The image is credited to St. Petersburg State University.
Original Research: Open access
“RNA-binding protein FXR1 is presented in rat brain in amyloid form”. Julia V. Sopova, Elena I. Koshel, Tatiana A. Belashova, Sergey P. Zadorsky, Alexandra V. Sergeeva, Vera A. Siniukova, Alexandr A. Shenfeld, Maria E. Velizhanina, Kirill V. Volkov, Anton A. Nizhnikov, Daniel V. Kachkin, Elena R. Gaginskaya & Alexey P. Galkin.
Scientific Reports doi:10.1038/s41598-019-55528-6.
Abstract
RNA-binding protein FXR1 is presented in rat brain in amyloid form
Amyloids are β-sheets-rich protein fibrils that cause neurodegenerative and other incurable human diseases affecting millions of people worldwide. However, a number of proteins is functional in the amyloid state in various organisms from bacteria to humans. Using an original proteomic approach, we identified a set of proteins forming amyloid-like aggregates in the brain of young healthy rats. One of them is the FXR1 protein, which is known to regulate memory and emotions. We showed that FXR1 clearly colocalizes in cortical neurons with amyloid-specific dyes Congo-Red, Thioflavines S and T. FXR1 extracted from brain by immunoprecipitation shows yellow-green birefringence after staining with Congo red. This protein forms in brain detergent-resistant amyloid oligomers and insoluble aggregates. RNA molecules that are colocalized with FXR1 in cortical neurons are insensitive to treatment with RNase A. All these data suggest that FXR1 functions in rat brain in amyloid form. The N-terminal amyloid-forming fragment of FXR1 is highly conserved across mammals. We assume that the FXR1 protein may be presented in amyloid form in brain of different species of mammals, including humans.






