Summary: Researchers have identified a new target of alcohol in the brain. A new study reveals alcohol blocks the KCNK13 potassium channel within the membrane of dopamine releasing neurons in the ventral tegmental area.
Source: University of Illinois.
When alcohol enters the brain, it causes neurons in a specialized region called the ventral tegmental area, or VTA — also known as the “pleasure center” — to release dopamine, a neurotransmitter that produces those feel-good sensations, and tells the brain that whatever it just experienced is worth getting more of.
Scientists have long sought the first step in the molecular pathway by which alcohol causes neurons in the VTA to release dopamine.
Now, researchers in the Center for Alcohol Research in Epigenetics at the University of Illinois at Chicago report in the journal Neuropharmacology, that alcohol blocks a potassium channel called KCNK13 that sits within the membrane of dopamine-releasing neurons in the VTA. When the potassium channel gets blocked, the neurons increase their activity and release more dopamine.
“The KCNK13 channel is absolutely required for alcohol to stimulate the release of dopamine by these neurons,” said Mark Brodie, professor of physiology and biophysics in the UIC College of Medicine and lead author of the study. “Without the channel, alcohol can’t stimulate the release of dopamine, and so drinking is likely less rewarding. We think that the KCNK13 channel presents an extremely exciting new target for drugs that could potentially help people with alcohol use disorder to stop drinking.”
Other drugs on the market to treat alcohol use disorder cause feelings of nausea with drinking, or interfere with the action of alcohol in other parts of the brain.
“Currently available drugs reduce the impact of alcohol on the brain that is akin to turning down the volume on a stereo,” he said. “A drug that would target KCNK13 would be different in that it would be like an on/off switch. If it’s turned off, alcohol just wouldn’t trigger increased dopamine release.”
Brodie explained that without the channel, the VTA would still be able to release dopamine in response to other pleasurable indulgences, like chocolate cake.
“This channel seems to be specific to alcohol effects in the VTA, so targeting it with a drug would dampen the effects of alcohol only,” he said.
Brodie and his colleagues used genetic techniques to reduce KCNK13 in the VTA of mice by about 15 percent compared with normal mice. When allowed to binge on alcohol, these mice drank 20 percent to 30 percent more than normal mice.
“We believe that mice with less KCNK13 in the VTA drank more alcohol in order to achieve the same ‘reward’ from alcohol as normal mice, presumably because alcohol was triggering the release of less dopamine in their brains,” Brodie said.
In another experiment, the researchers examined the response of neurons in the VTA region taken from the mice that expressed less KCNK13. When these neurons were exposed to alcohol, they were 50 percent less responsive to alcohol than VTA neurons from normal mice.
Brodie speculates that variations in the amount of the KCNK13 channel could be involved in predisposing certain people to binge drinking.
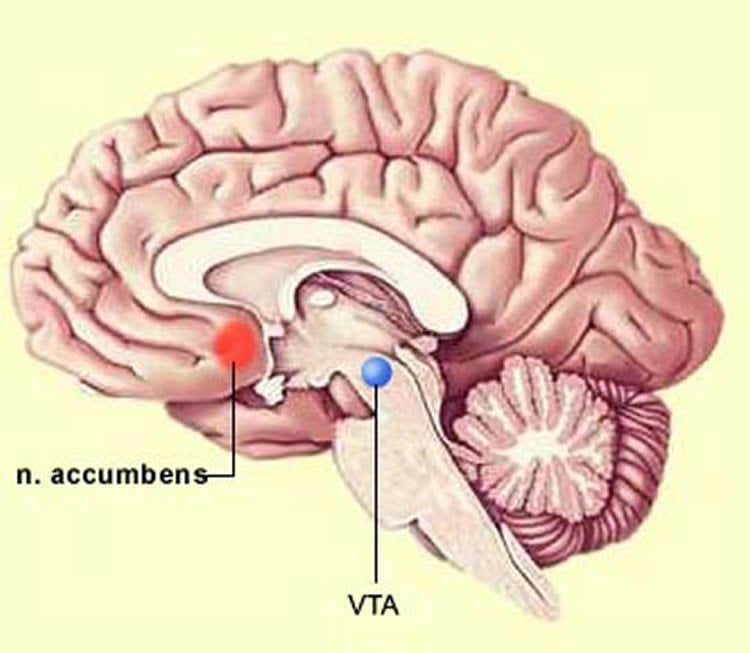
“If someone has naturally lower levels of this channel, then in order to produce the pleasurable effects of alcohol, that person would have to drink much more, and may be at higher risk for binge drinking disorder,” he said.
Brodie and his colleagues will continue to investigate the role of KCNK13 and examine how selective manipulation of the channel in other brain areas and cell types might alter alcohol-related behaviors.
“We are the first to show that KCNK13 is a primary, direct target of alcohol and that this channel is important for regulating alcohol consumption. KCNK13 represents a novel target for the development of alcohol use disorder drugs, of which we have relatively few today,” Brodie said.
Funding: This research was funded by Grant R01AA05846 and P50AA022538 from the National Institute on Alcohol Abuse and Alcoholism of the National Institutes of Health.
Chang You, Bertha Vandegrift, Donghong He, Subhash Pandey and Amy Lasek of the University of Illinois at Chicago and Antonia Savarese (currently at Oregon Health and Science University) are co-authors on the paper.
Source: Sharon Parmet – University of Illinois
Publisher: Organized by NeuroscienceNews.com.
Image Source: NeuroscienceNews.com image is in the public domain.
Original Research: Open access research for “Ethanol acts on KCNK13 potassium channels in the ventral tegmental area to increase firing rate and modulate binge–like drinking” by Chang You, Antonia Savarese, Bertha J. Vandegrift, Donghong He, Subhash C. Pandey, Amy W. Lasek, Mark S. Brodie in Neuropharmacology. Published October 24 2018.
doi:10.1016/j.neuropharm.2018.10.008
[cbtabs][cbtab title=”MLA”]University of Illinois”New Target of Alcohol in the Brain Identified.” NeuroscienceNews. NeuroscienceNews, 24 October 2018.
<https://neurosciencenews.com/alcohol-brain-target-10085/>.[/cbtab][cbtab title=”APA”]University of Illinois(2018, October 24). New Target of Alcohol in the Brain Identified. NeuroscienceNews. Retrieved October 24, 2018 from https://neurosciencenews.com/alcohol-brain-target-10085/[/cbtab][cbtab title=”Chicago”]University of Illinois”New Target of Alcohol in the Brain Identified.” https://neurosciencenews.com/alcohol-brain-target-10085/ (accessed October 24, 2018).[/cbtab][/cbtabs]
Abstract
Ethanol acts on KCNK13 potassium channels in the ventral tegmental area to increase firing rate and modulate binge–like drinking
Alcohol excitation of the ventral tegmental area (VTA) is important in neurobiological processes related to the development of alcoholism. The ionotropic receptors on VTA neurons that mediate ethanol-induced excitation have not been identified. Quinidine blocks ethanol excitation of VTA neurons, and blockade of two-pore potassium channels is among the actions of quinidine. Therefore two-pore potassium channels in the VTA may be potential targets for the action of ethanol. Here, we explored whether ethanol activation of VTA neurons is mediated by the two-pore potassium channel KCNK13. Extracellular recordings of the response of VTA neurons to ethanol were performed in combination with knockdown of Kcnk13 using a short hairpin RNA (shRNA) in C57BL/6 J mice. Real-time PCR and immunohistochemistry were used to examine expression of this channel in the VTA. Finally, the role of KCNK13 in binge-like drinking was examined in the drinking in the dark test after knockdown of the channel. Kcnk13 expression in the VTA was increased by acute ethanol exposure. Ethanol-induced excitation of VTA neurons was selectively reduced by shRNA targeting Kcnk13. Importantly, knockdown of Kcnk13 in the VTA resulted in increased alcohol drinking. These results are consistent with the idea that ethanol stimulates VTA neurons at least in part by inhibiting KCNK13, a specific two-pore potassium channel, and that KCNK13 can control both VTA neuronal activity and binge drinking. KCNK13 is a novel alcohol-sensitive molecular target and may be amenable to the development of pharmacotherapies for alcoholism treatment.