Summary: Researchers have been able to provide a crystal structure of the extracellular domain of ZIP4.
Source: Michigan State University.
Mapping the molecular structure where medicine goes to work is a crucial step toward drug discovery against deadly diseases.
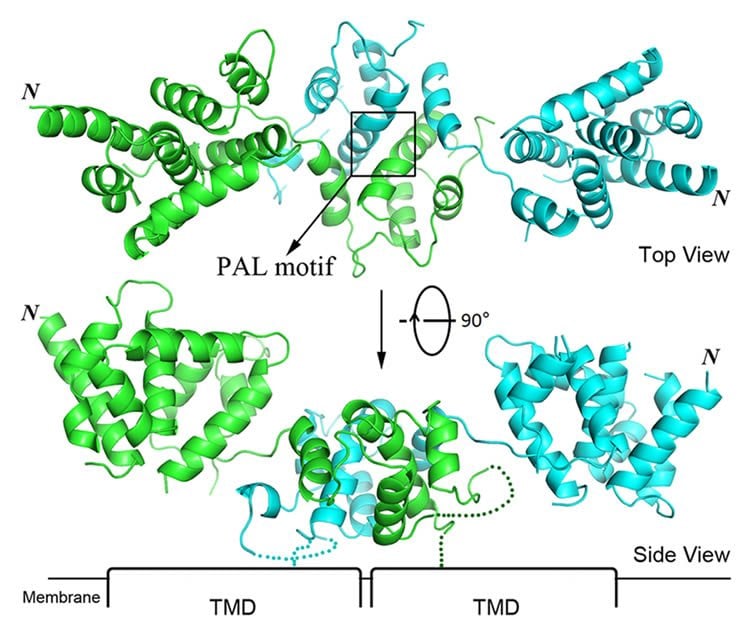
Researchers at Michigan State University have taken that critical first step by providing a crystal structure of the extracellular domain, or ECD, of ZIP4 – the exclusive protein responsible for the uptake of zinc from food. The ZIP family consists of thousands of zinc/iron transporter proteins, and this work represents the first-ever structural information of the ZIP family at the atomic level.
The results are published in the current issue of Nature Communications and provide a roadmap for potential target sites for people suffering from acrodermitis enteropathica, a rare but lethal genetic disorder leading to severe zinc deficiency, and pancreatic cancer where ZIP4 is abnormally overexpressed.
“Many drug candidates fail during development because their targets are buried inside the cell,” said Jian Hu, assistant professor in MSU’s chemistry department. “With ZIP4, though, the large ECD is fully exposed to the extracellular space and quite accessible.”
Hu revealed that ZIP4-ECD acts as a critical accessory domain that is essential for optimal zinc transport. Therefore, targeting it appears to be a promising strategy regulating the function of ZIP4.
The study also revealed that many human ZIP proteins share a common architecture in their ECDs. This sheds light on structural and functional studies of other ZIP proteins involved in a variety of cancers, osteoarthritis and other serious diseases. Thanks to Hu’s lab, scientists now have a research foundation on which to further study zinc transport mechanism of ZIP proteins.
Hu was drawn to study zinc and other trace elements because they are essential for life, and zinc is the second most-common trace element behind iron. He focuses on deciphering how the body maintains proper levels. He also dedicates his efforts to exploring what happens when amounts of trace elements rise to toxic levels.
“For example, for patients suffering from diseases like Alzheimer’s or Parkinson’s, the levels of transition metals, particularly zinc and iron, in their brains are significantly higher than those of healthy people,” Hu said. “My laboratory is interested in revealing a better understanding of the body’s system of properly handling these trace elements.”
Finding: This research was funded in part by the National Institutes of Health.
Source: Gerry Everding – Michigan State University
Image Source: This NeuroscienceNews.com image is credited to Jian Hu.
Original Research: Full open access research for “Structural insights of ZIP4 extracellular domain critical for optimal zinc transport” by Tuo Zhang, Dexin Sui and Jian Hu in Nature Communications . Published online June 20 2016 doi:10.1038/ncomms11979
[cbtabs][cbtab title=”MLA”]Michigan State University. “Molecular Map Provides Clues to Zinc Related Diseases.” NeuroscienceNews. NeuroscienceNews, 20 June 2016.
<https://neurosciencenews.com/zinc-disease-genetics-4530/>.[/cbtab][cbtab title=”APA”]Michigan State University. (2016, June 20). Molecular Map Provides Clues to Zinc Related Diseases. NeuroscienceNew. Retrieved June 20, 2016 from https://neurosciencenews.com/zinc-disease-genetics-4530/[/cbtab][cbtab title=”Chicago”]Michigan State University. “Molecular Map Provides Clues to Zinc Related Diseases.” https://neurosciencenews.com/zinc-disease-genetics-4530/ (accessed June 20, 2016).[/cbtab][/cbtabs]
Abstract
Structural insights of ZIP4 extracellular domain critical for optimal zinc transport
The ZIP zinc transporter family is responsible for zinc uptake from the extracellular milieu or intracellular vesicles. The LIV-1 subfamily, containing nine out of the 14 human ZIP proteins, is featured with a large extracellular domain (ECD). The critical role of the ECD is manifested by disease-causing mutations on ZIP4, a representative LIV-1 protein. Here we report the first crystal structure of a mammalian ZIP4-ECD, which reveals two structurally independent subdomains and an unprecedented dimer centred at the signature PAL motif. Structure-guided mutagenesis, cell-based zinc uptake assays and mapping of the disease-causing mutations indicate that the two subdomains play pivotal but distinct roles and that the bridging region connecting them is particularly important for ZIP4 function. These findings lead to working hypotheses on how ZIP4-ECD exerts critical functions in zinc transport. The conserved dimeric architecture in ZIP4-ECD is also demonstrated to be a common structural feature among the LIV-1 proteins.
“Structural insights of ZIP4 extracellular domain critical for optimal zinc transport” by Tuo Zhang, Dexin Sui and Jian Hu in Nature Communications . Published online June 20 2016 doi:10.1038/ncomms11979