Summary: A protein called Tmep appears to control neuron excitability by altering the behavior of an ion channel, impacting the likelihood of whether that neuron will fire.
Source: University of Arizona
In many cases of epilepsy—a disorder that disrupts the normal pattern of electrical activity in the brain and often results in seizures—the underlying cause is unknown, according to the National Institute of Neurological Disorders and Stroke.
Researchers at the University of Arizona might have found an explanation for some forms of epilepsy. Their results are published in Molecular Neurobiology.
The researchers studied a gene in mice that influences the formation and function of connections between muscle and motor neurons, which control bodily movement.
They discovered that when a protein called TMEM184B, which is found on cell membranes of neurons, isn’t present, neurons appeared damaged and fired too often, said lead study author Tiffany Cho, who was a research technician in the UArizona Bhattacharya Lab when the research was conducted. Cho will start as a medical student at Liberty University Medical School in Virginia in the fall.
The researchers wondered how the seemingly damaged neurons might affect the ability of the neuron to fire the muscles properly. So, Cho and her co-authors next investigated the corresponding protein, called Tmep, in fruit flies, which are easier to study at the cellular level.
“What we found in the fruit flies was that the neurons seem to overreact to an individual stimulus,” said study-coauthor Martha Bhattacharya, an assistant professor in the Department of Neuroscience and director of the Bhattacharya Lab, which explores early stages of deterioration of brain cell health in neurodegenerative diseases.
This suggests that Tmep—and TMEM184B by extension—are responsible for controlling excitability of neurons.
“This is related to what happens to patients with epilepsy, so we think we may have identified a gene involved in some forms of epilepsy that don’t have another explanation,” Bhattacharya said.
The researchers think that Tmep may alter the behavior of ion channels, which control the amount of calcium in the cell, and therefore the likelihood that the neuron will fire.
“When we saw cellular changes in the fruit flies, it really made us think about whether this was also controlling balance of ions, for example charged particles such as calcium in neurons because that’s a common thing that happens with epilepsy. No one had looked at whether this protein controls ion levels until this paper,” Bhattacharya said.
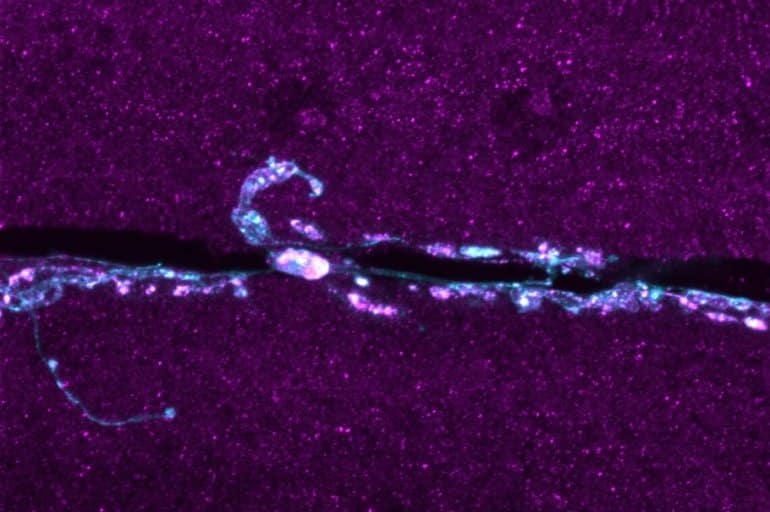
Bhattacharya has also connected with clinicians who have done gene sequencing of alterations of the TMEM184B protein in humans.
“One of the things we’d like to figure out is if those mutations, especially the ones in which the patients have epilepsy or something related, cause this overexcitability that we see,” Bhattacharya said.
“We can do that in the fruit fly model because we have ways of measuring electrical activity, and that’s something we did in the paper. What we’re trying to do is put those human mutations into the fly genome and see if they cause the same changes in the excitability of the neurons. And if they do that, then we want to know why.”
The researchers also noticed that the fly larvae without Tmep moved much more slowly than other flies when crawling across a plate. As a result, they are curious to see if the protein might also play a role in other neuromuscular diseases—such as amyotrophic lateral sclerosis, or ALS—in addition to disorders like epilepsy.
About this genetics and epilepsy research news
Author: Mikayla Mace Kelley
Source: University of Arizona
Contact: Mikayla Mace Kelley – University of Arizona
Image: The image is credited to Martha Bhattacharya
Original Research: Closed access.
“The Putative Drosophila TMEM184B Ortholog Tmep Ensures Proper Locomotion by Restraining Ectopic Firing at the Neuromuscular Junction” by Tiffany S. Cho et al. Molecular Neurobiology
Abstract
The Putative Drosophila TMEM184B Ortholog Tmep Ensures Proper Locomotion by Restraining Ectopic Firing at the Neuromuscular Junction
TMEM184B is a putative seven-pass membrane protein that promotes axon degeneration after injury. TMEM184B mutation causes aberrant neuromuscular architecture and sensory and motor behavioral defects in mice. The mechanism through which TMEM184B causes neuromuscular defects is unknown.
We employed Drosophila melanogaster to investigate the function of the closely related gene, Tmep (CG12004), at the neuromuscular junction. We show that Tmep is required for full adult viability and efficient larval locomotion. Tmep mutant larvae have a reduced body contraction rate compared to controls, with stronger deficits in females.
In recordings from body wall muscles, Tmep mutants show substantial hyperexcitability, with many postsynaptic potentials fired in response to a single stimulation, consistent with a role for Tmep in restraining synaptic excitability.
Additional branches and satellite boutons at Tmep mutant neuromuscular junctions are consistent with an activity-dependent synaptic overgrowth. Tmep is expressed in endosomes and synaptic vesicles within motor neurons, suggesting a possible role in synaptic membrane trafficking.
Using RNAi knockdown, we show that Tmep is required in motor neurons for proper larval locomotion and excitability, and that its reduction increases levels of presynaptic calcium. Locomotor defects can be rescued by presynaptic knockdown of endoplasmic reticulum calcium channels or by reducing evoked release probability, further suggesting that excess synaptic activity drives behavioral deficiencies.
Our work establishes a critical function for Tmep in the regulation of synaptic transmission and locomotor behavior.






