Summary: Using cryo electron microscopy, researchers have successfully images serotonin attaching to the receptor and twisting open the channel, allowing sodium molecules to travel through.
Source: Case Western Reserve University.
Serotonin (3A) receptors are common drug targets in the treatment of pain, gastrointestinal dysfunctions, and mood disorders yet little is known about their three-dimensional structure. Details about serotonin receptor structures could provide important clues to designing better drugs with less side effects. Now, a team of researchers from Case Western Reserve University School of Medicine have used high-powered microscopes to view serotonin activating its receptor for the first time. Images published in Nature reveal molecular details about the receptor that could improve drug design to treat a multitude of diseases.
Serotonin receptors sit in cell membranes throughout the body, including the brain, stomach, and the associated nervous system. Drugs that inhibit serotonin receptors help control post-operative nausea, support cancer therapies, and are used to treat gastrointestinal conditions like irritable bowel syndrome. These inhibitors also find application as anti-depressants, and promote attention and memory.
Broad application comes with side effects–in part due to suboptimal drug-receptor interactions, says study lead Sudha Chakrapani, PhD, associate professor of physiology and biophysics at Case Western Reserve University School of Medicine. “Successful design of safer therapeutics has been slowed because there is a limited understanding of the structure of the serotonin receptor itself, and what happens after serotonin binds to it. Our work is the first to describe how serotonin activates the full-length serotonin receptor at a level of detail to nearly the individual atom.”
Using Nobel Prize-winning microscope technology, Chakrapani’s team studied serotonin as it interacted with its receptor. Their images show serotonin attaching to the receptor and twisting open the channel. Open channels allow molecules to travel from outside the cell to inside. The researchers used simulations to watch as sodium molecules traveled through newly opened channels. The new study highlights distinct conformations of the serotonin receptor that would make a cell more or less permeable to certain molecules–a key insight for drug developers. It also shows which portions of the receptor are most critical for proper channel function.
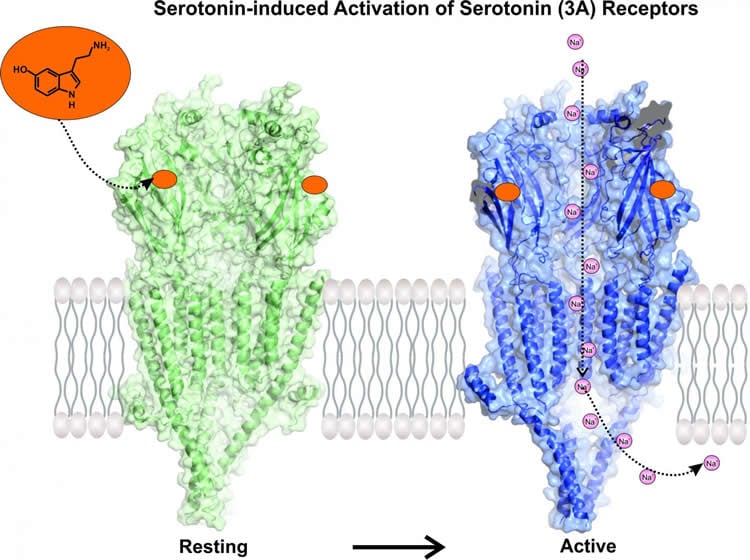
The entire serotonin-receptor occurs in a space about a few billionths of a meter across. Microscopes have only recently evolved to capture such tiny molecules. Cutting-edge technology in the new study–cryo-electron microscopy–earned a 2017 Nobel Prize in chemistry. It uses high-powered microscopes to take snapshots of proteins in action, and compiles them into three-dimensional structural models. In the past year it has already helped Case Western Reserve researchers view structures for proteins central to kidney stones and other ailments. Chakrapani used “cryo-EM” last year to view the serotonin receptor alone, laying the foundation for the present study.
The researchers hope their findings could lead to more precise drugs that target specific regions or functions of serotonin receptors. “It’s likely that new and different drugs can work as effective serotonin inhibitors, especially if they’re designed to work differently than current drugs,” says first author Sandip Basak, PhD, postdoctoral fellow in Chakrapani’s lab. “We’re actively pursuing these approaches to help design safer therapeutics that modulate the serotonin receptor to treat a range of conditions.”
Funding: This research was, in part, supported by the National Cancer Institute’s National Cryo-EM Facility at the Frederick National Laboratory for Cancer Research. The researchers also received assistance from the Cleveland Center for Membrane and Structural Biology and Department of Ophthalmology and Visual Sciences (NIH Core Grant P30EY11373). The study was also supported by a National Institutes of Health grant (1R01GM108921), and Cryo-EM supplement (3R01GM108921-03S1) to S.C and the American Heart Association postdoctoral Fellowship to S.B (17POST33671152).
Co-authors Mark Sansom, PhD, professor of biochemistry at University of Oxford and Shanlin Rao, a graduate student in his lab, carried out molecular dynamics simulations on the different conformational states of the serotonin receptors for this study.
Source: Ansley Gogol – Case Western Reserve University
Publisher: Organized by NeuroscienceNews.com.
Image Source: NeuroscienceNews.com image is credited to Case Western Reserve University School of Medicine.
Original Research: Abstract for “Cryo-EM reveals two distinct serotonin-bound conformations of full-length 5-HT3A receptor” by Sandip Basak, Yvonne Gicheru, Shanlin Rao, Mark S. P. Sansom & Sudha Chakrapani in Nature. Published October 31 2018.
doi:10.1038/s41586-018-0660-7
[cbtabs][cbtab title=”MLA”]Case Western Reserve University”New Images Show Serotonin Activating its Receptor for First Time.” NeuroscienceNews. NeuroscienceNews, 2 November 2018.
<https://neurosciencenews.com/serotonin-receptor-activation-10137/>.[/cbtab][cbtab title=”APA”]Case Western Reserve University(2018, November 2). New Images Show Serotonin Activating its Receptor for First Time. NeuroscienceNews. Retrieved November 2, 2018 from https://neurosciencenews.com/serotonin-receptor-activation-10137/[/cbtab][cbtab title=”Chicago”]Case Western Reserve University”New Images Show Serotonin Activating its Receptor for First Time.” https://neurosciencenews.com/serotonin-receptor-activation-10137/ (accessed November 2, 2018).[/cbtab][/cbtabs]
Abstract
Cryo-EM reveals two distinct serotonin-bound conformations of full-length 5-HT3A receptor
The 5-HT3A serotonin receptor1, a cationic pentameric ligand-gated ion channel (pLGIC), is the clinical target for management of nausea and vomiting associated with radiation and chemotherapies2. Upon binding, serotonin induces a global conformational change that encompasses the ligand-binding extracellular domain (ECD), the transmembrane domain (TMD) and the intracellular domain (ICD), the molecular details of which are unclear. Here we present two serotonin-bound structures of the full-length 5-HT3A receptor in distinct conformations at 3.32 Å and 3.89 Å resolution that reveal the mechanism underlying channel activation. In comparison to the apo 5-HT3A receptor, serotonin-bound states underwent a large twisting motion in the ECD and TMD, leading to the opening of a 165 Å permeation pathway. Notably, this motion results in the creation of lateral portals for ion permeation at the interface of the TMD and ICD. Combined with molecular dynamics simulations, these structures provide novel insights into conformational coupling across domains and functional modulation.






