Summary: According to researchers, REM sleep is suppressed by adenosine acting on a specific type of subreceptor in the olfactory bulb.
Source: University of Tsukuba.
The regulation and function of sleep is one of the biggest black boxes of today’s brain science. A new paper published online on August 2 in the journal Brain Structure & Function finds that rapid eye movement (REM) sleep is suppressed by adenosine acting on a specific subtype of adenosine receptors, the A2A receptors, in the olfactory bulb.
The study was conducted by researchers at Fudan University’s School of Basic Medical Sciences in the Department of Pharmacology and the University of Tsukuba’s International Institute for Integrative Sleep Medicine (WPI-IIIS). The research team used pharmacological and genetic methods to show that blocking A2A receptors or neurons that contain the A2A receptors in the olfactory bulb increases REM sleep in rodents.
Adenosine has long been known to represent a state of relative energy deficiency and to induce sleep by blocking wakefulness. The new findings demonstrate for the first time that adenosine also inhibits REM sleep, a unique phase of sleep in mammalians that is characterized by random eye movement and low muscle tone throughout the body. The Chinese-Japanese research team discovered that adenosine acts specifically in the olfactory bulb which transmits odor information from the nose to the brain. Because olfactory dysfunction can be treated with an A2A receptor antagonist, for example caffeine, it is possible that REM sleep and the perception of odors are linked in the olfactory bulb. Interestingly, the ability to smell is reduced in patients with REM sleep behavior disorder (RBD). Dreams which mostly occur during REM sleep are usually a pure mental activity while the body is at rest. However, patients who suffer from RBD act out their dreams.
Yiqun Wang, the lead investigator on this project said that “our findings encourage us to believe that A2A receptors may be a novel target to treat RBD by suppressing REM sleep. Our observation clearly suggest an intriguing possibility for treating this disease with an A2A receptor agonist or allosteric modulator.”
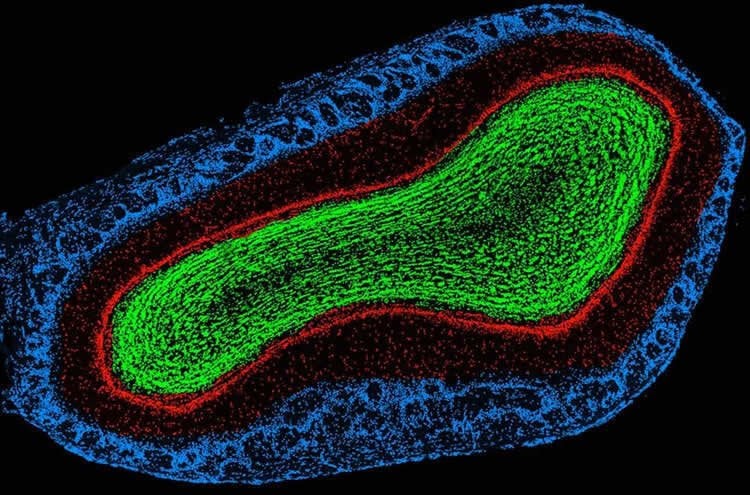
The Department of Pharmacology in the School of Basic Medical Sciences at Fudan University is focusing on molecular mechanisms of sleep initiation and maintenance, neural circuits of sleep/wake regulation and effects of sleep on advanced cognitive function. These researches are aimed to promote the understanding of the sleep process, strengthen the prevention and treatment of sleep related diseases and improve the national health and social productivity.
WPI-IIIS was launched by the Ministry of Education, Culture, Sports, Science and Technology of Japan with the aim of building globally visible research centers. At WPI-IIIS gather globally prominent scientists from multiple research fields contributing to elucidate the fundamental principles of sleep/wake regulation, and develop new strategies to assess and treat sleep diseases as well as the closely associated metabolic and mental disorders.
Source: Masataka Sasabe – University of Tsukuba
Image Source: This NeuroscienceNews.com image is in the public domain.
Original Research: Abstract for “Adenosine A2A receptors in the olfactory bulb suppress rapid eye movement sleep in rodents” by Yi-Qun Wang, Rui Li, Dian-Ru Wang, Yoan Cherasse, Ze Zhang, Meng-Qi Zhang, Oriana Lavielle, Kristopher McEown, Serge N. Schiffmann, Alban de Kerchove d’Exaerde, Wei-Min Qu, Michael Lazarus, and Zhi-Li Huang in Brain Structure & Function. Published online August 2 2016 doi:10.1007/s00429-016-1281-2
[cbtabs][cbtab title=”MLA”]University of Tsukuba. “New Role of Adenosine in the Regulation of REM Sleep Discovered.” NeuroscienceNews. NeuroscienceNews, 1 September 2016.
<https://neurosciencenews.com/rem-sleep-adenosine-4942/>.[/cbtab][cbtab title=”APA”]University of Tsukuba. (2016, September 1). New Role of Adenosine in the Regulation of REM Sleep Discovered. NeuroscienceNews. Retrieved September 1, 2016 from https://neurosciencenews.com/rem-sleep-adenosine-4942/[/cbtab][cbtab title=”Chicago”]University of Tsukuba. “New Role of Adenosine in the Regulation of REM Sleep Discovered.” https://neurosciencenews.com/rem-sleep-adenosine-4942/ (accessed September 1, 2016).[/cbtab][/cbtabs]
Abstract
Adenosine A2A receptors in the olfactory bulb suppress rapid eye movement sleep in rodents
Rapid eye movement (REM) sleep behavior disorder in humans is often accompanied by a reduced ability to smell and detect odors, and olfactory bulbectomized rats exhibit increased REM sleep, suggesting that the olfactory bulb (OB) is involved in REM-sleep regulation. However, the molecular mechanism of REM-sleep regulation by the OB is unknown. Adenosine promotes sleep and its A2A receptors (A2AR) are expressed in the OB. We hypothesized that A2AR in the OB regulate REM sleep. Bilateral microinjections of the A2AR antagonist SCH58261 into the rat OB increased REM sleep, whereas microinjections of the A2AR agonist CGS21680 decreased REM sleep. Similar to the A2AR antagonist, selective A2AR knockdown by adeno-associated virus carrying short-hairpin RNA for A2AR in the rat OB increased REM sleep. Using chemogenetics on the basis of designer receptors exclusively activated by designer drugs, we demonstrated that the inhibition of A2AR neurons increased REM sleep, whereas the activation of these neurons decreased REM sleep. Moreover, using a conditional anterograde axonal tract-tracing approach, we found that OB A2AR neurons innervate the piriform cortex and olfactory tubercle. These novel findings indicate that adenosine suppresses REM sleep via A2AR in the OB of rodents.
“Adenosine A2A receptors in the olfactory bulb suppress rapid eye movement sleep in rodents” by Yi-Qun Wang, Rui Li, Dian-Ru Wang, Yoan Cherasse, Ze Zhang, Meng-Qi Zhang, Oriana Lavielle, Kristopher McEown, Serge N. Schiffmann, Alban de Kerchove d’Exaerde, Wei-Min Qu, Michael Lazarus, and Zhi-Li Huang in Brain Structure & Function. Published online August 2 2016 doi:10.1007/s00429-016-1281-2