Summary: The origin of the axon differs between primates and non-primates in the architecture of neurons, a new study reports.
Source: RUB
Researchers from the research group Developmental Neurobiology at Ruhr-Universität Bochum around Professor Petra Wahle, in collaboration with partners from Mannheim and Jülich, Germany, and Linz, Austria, and La Laguna, Spain, have shown that primates and non-primates differ in an important aspect of their architecture: the origin of the axon, which is the process responsible for the transmission of electrical signals called action potentials.
Results were published 20 April 2022 in the Journal eLife.
Axons can emerge from dendrites
Until now, it was considered textbook knowledge that the axon always, with few exceptions, arises from the cell body of a neuron. However, it may also originate from dendrites, which serve to collect and integrate the incoming synaptic signals. These are called axon-carrying dendrites.
“A unique aspect of the project is that the team worked with archived tissue and slide preparations, which included material that has been used for years to teach students,” explains Petra Wahle.
In addition, a range of species was studied, including rodents (mouse, rat), ungulates (pig), carnivores (cat, ferret), and macaque and human of the zoological order primates. The use of five staining methods and assessment of more than 34,000 neurons led the group to conclude that there is a species difference between non-primates and primates.
Excitatory pyramidal neurons in particular of the outer layers II and III of the cerebral cortex of primates has clearly fewer axon-carrying dendrites than pyramidal neurons of non-primates.
Further, quantitative differences in the proportion of axon-carrying dendrite cells were found within the species cat and human for inhibitory interneurons. No quantitative differences were observed when comparing in macaque cortical areas with primary sensory and higher brain functions.
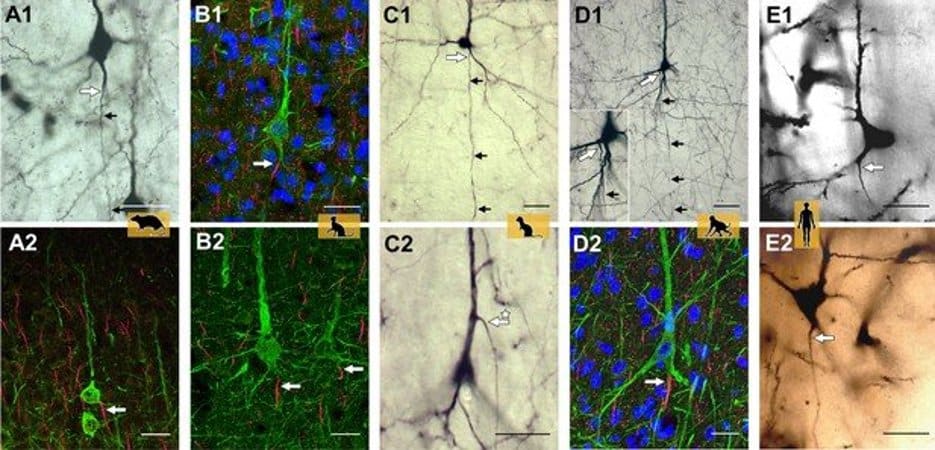
High-resolution microscopy was of particular importance, as Petra Wahle describes: “This allowed the detection of axonal origins accurately tracked at the micrometer level, which is sometimes not so easy with conventional light microscopy.”
Evolutionary advantage still enigmatic
Little is known on the function of axon-carrying dendrites. Usually, a neuron integrates excitatory inputs arriving at the dendrites with inhibitory inputs, a process termed somatodendritic integration.
The neuron then decides if inputs are strong enough and important enough to be transmitted via action potentials to other neurons and brain areas. Axon-carrying dendrites are considered privileged because depolarizing inputs to these dendrites are able to evoke action potentials directly without involvement of somatic integration and somatic inhibition.
Why this species difference has evolved, and the potential advantage it may have for the neocortical information processing in primates, is as yet unknown.
About this evolutionary neuroscience research news
Author: Press Office
Source: RUB
Contact: Press Office – RUB
Image: The image is credited to the researchers
Original Research: Open access.
“Neocortical pyramidal neurons with axons emerging from dendrites are frequent in non-primates, but rare in monkey and human” by Petra Wahle et al. eLife
Abstract
Neocortical pyramidal neurons with axons emerging from dendrites are frequent in non-primates, but rare in monkey and human
The canonical view of neuronal function is that inputs are received by dendrites and somata, become integrated in the somatodendritic compartment and upon reaching a sufficient threshold, generate axonal output with axons emerging from the cell body. The latter is not necessarily the case. Instead, axons may originate from dendrites.
The terms ‘axon carrying dendrite’ (AcD) and ‘AcD neurons’ have been coined to describe this feature. In rodent hippocampus, AcD cells are shown to be functionally ‘privileged’, since inputs here can circumvent somatic integration and lead to immediate action potential initiation in the axon.
Here, we report on the diversity of axon origins in neocortical pyramidal cells of rodent, ungulate, carnivore, and primate.
Detection methods were Thy-1-EGFP labeling in mouse, retrograde biocytin tracing in rat, cat, ferret, and macaque, SMI-32/βIV-spectrin immunofluorescence in pig, cat, and macaque, and Golgi staining in macaque and human. We found that in non-primate mammals, 10–21% of pyramidal cells of layers II–VI had an AcD. In marked contrast, in macaque and human, this proportion was lower and was particularly low for supragranular neurons.
A comparison of six cortical areas (being sensory, association, and limbic in nature) in three macaques yielded percentages of AcD cells which varied by a factor of 2 between the areas and between the individuals. Unexpectedly, pyramidal cells in the white matter of postnatal cat and aged human cortex exhibit AcDs to much higher percentages.
In addition, interneurons assessed in developing cat and adult human cortex had AcDs at type-specific proportions and for some types at much higher percentages than pyramidal cells.
Our findings expand the current knowledge regarding the distribution and proportion of AcD cells in neocortex of non-primate taxa, which strikingly differ from primates where these cells are mainly found in deeper layers and white matter.







