Summary: Researchers have identified an organelle within neurons we use to perceive different odors.
Source: Umea University
Researchers at Umeå University have discovered a previously unknown cellular component, an organelle, inside neurons that we use to perceive smell.
The discovery may have implications for further research on impaired sense of smell, which is a common symptom of COVID-19.
“A prerequisite for finding a treatment for impaired sense of smell is to first understand how the sense of smell works, says Staffan Bohm,” Professor at the Department of Molecular Biology at Umeå University.
What the researchers have discovered is a so-called organelle inside nerve cells, that has not previously been observed. The newly discovered organelle has been given the name multivesicular transducosome by the researchers. The discovery was made possible thanks to Umeå University’s unique microscopy infrastructure.
Organelles are distinct “workstations” inside cells that can be compared to the different organs of the body, i.e., different organelles have different functions in the cell. Most organelles are common to different cell types, but there are also organelles with specific functions that only occur in certain cell types.
Olfactory nerve cells have long projections, or cilia, that protrude into the nasal cavity and contain the proteins that bind odorous substances and thus initiate nerve impulses to the brain. The conversion of odor into nerve impulses is called transduction and the newly discovered organelle, contains only transduction proteins.
The role of the transductosome is to both store and keep transduction proteins separate from each other until they are needed. Upon olfactory stimulation, the outer membrane of the organelle ruptures, releasing the transduction proteins so that they can reach the cilia of the neuron, and smell is perceived.
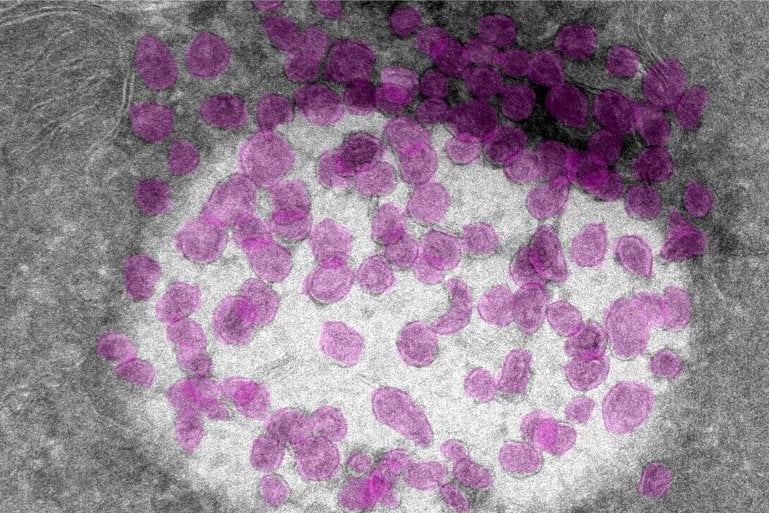
The researchers also discovered that the transductosome carries a protein called retinitis pigmentosa 2, RP2, which is otherwise known to regulate transduction in the eye’s photoreceptor cells. If the RP2 gene is mutated, it can cause a variant of the eye disease retinitis pigmentosa that damages the eye’s light-sensitive cells.
“A question for further research is whether the transductosome has a role in vision and whether it is present in brain neurons that are activated by neurotransmitters and not light and smell. If so, the discovery may prove even more significant,” says Staffan Bohm.
The transducosome was discovered when researcher Devendra Kumar Maurya used a new technique called correlative microscopy. The technique combines electron microscopy and confocal microscopy so that a cell’s internal structures and the location of different proteins can be imaged simultaneously.
Crucial to the discovery was Devendra’s method development, which enabled the technique to be used to analyze intact neurons in tissue sections.
About this olfaction research news
Author: Press Office
Source: Umea University
Contact: Press Office – Umea University
Image: The image is credited to Devendra Kumar Maurya
Original Research: Open access.
“A multivesicular body-like organelle mediates stimulus-regulated trafficking of olfactory ciliary transduction proteins” by Devendra Kumar Maurya et al. Nature Communications
Abstract
A multivesicular body-like organelle mediates stimulus-regulated trafficking of olfactory ciliary transduction proteins
Stimulus transduction in cilia of olfactory sensory neurons is mediated by odorant receptors, Gαolf, adenylate cyclase-3, cyclic nucleotide-gated and chloride ion channels. Mechanisms regulating trafficking and localization of these proteins in the dendrite are unknown.
By lectin/immunofluorescence staining and in vivo correlative light-electron microscopy (CLEM), we identify a retinitis pigmentosa-2 (RP2), ESCRT-0 and synaptophysin-containing multivesicular organelle that is not part of generic recycling/degradative/exosome pathways.
The organelle’s intraluminal vesicles contain the olfactory transduction proteins except for Golf subunits Gγ13 and Gβ1. Instead, Gβ1 colocalizes with RP2 on the organelle’s outer membrane.
The organelle accumulates in response to stimulus deprivation, while odor stimuli or adenylate cyclase activation cause outer membrane disintegration, release of intraluminal vesicles, and RP2/Gβ1 translocation to the base of olfactory cilia.
Together, these findings reveal the existence of a dendritic organelle that mediates both stimulus-regulated storage of olfactory ciliary transduction proteins and membrane-delimited sorting important for G protein heterotrimerization.






