Summary: A new technique that uses ultrafast fMRI is able to capture brain activity at sub-second levels. The technique allows for real-time monitoring of the brain under stimulation conditions.
Source: University of Queensland
Brain stimulation, such as Deep brain stimulation (DBS), is a powerful way to treat neurological and psychiatric disorders. While it has provided therapeutic benefit for sufferers of Parkinson’s, Alzheimer’s, and addiction for more than a decade, its underlying neural mechanism is not yet fully understood.
Researchers at the Queensland Brain Institute (QBI) are now one step closer to unravelling the mystery of brain activity to better understand this mechanism and potentially predict DBS outcomes.
The brain is a highly complex network of circuits organised hierarchically with wide-ranging connections. Connections go in different directions, forwards and backwards, and between neurons that are either excitatory – the accelerators of a response – or inhibitory – the brakes modifying a response.
“Say you want to move your hand – once that signal is initiated, we expect that the activity that follows depends on the brain’s neural networks,” Associate Professor Kai-Hsiang Chuang said.
“What we don’t fully understand is how or when these structural and functional components of the brain interact to eventually lead to the outcome of moving your hand.”
Functional MRI (fMRI) is the most popular technique used to study brain networks. fMRI tracks blood flow and oxygenation changes following neural activity, thereby indirectly measuring the functional connections being formed, and giving us an indication of where brain activity is propagating.
Brain activity, however, isn’t as simple as a signal travelling from area to area.
The team at the Chuang laboratory have developed a new ultrafast fMRI technique with a vastly increased temporal resolution, enabling them to capture the dynamics of brain activity at a sub-second level.
Associate Professor Chuang said the new technique had led to more comprehensive understanding of how and when the brain’s structural and functional connections interact.
“The first new discovery we made is that brain activity not only propagates through structural wiring but follows certain preferential circuits depending on their excitatory and inhibitory neuronal distribution,” he said.
“Communication between brain regions of similar cell types becomes more fluent, and the brain activity stronger.”
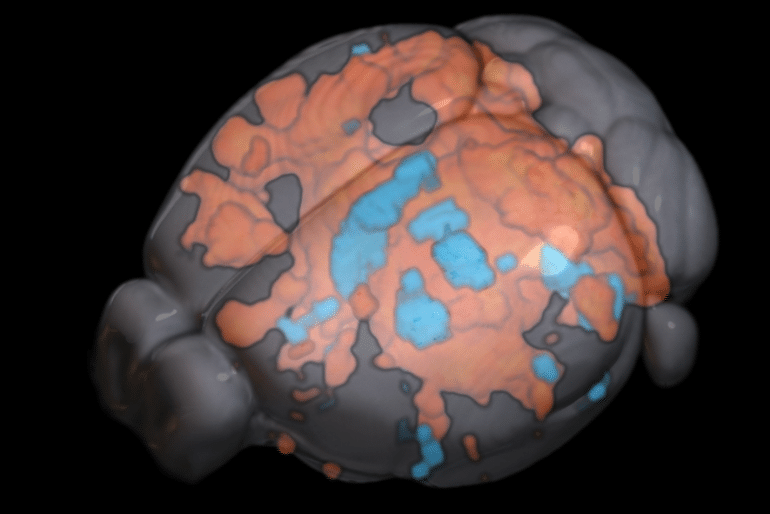
The Chuang group tracked the brain activity of mice both while stimulated and at rest using their ultrafast fMRI technique. When the brain was stimulated, activity followed the structural wiring in the forward direction — from A to B and then B to C. When the brain was at rest, activity was more dependent on cell type organisation and less on structural wiring, propagating between C and B but not with A, if that’s where the preferential circuit was.
This means that how information is processed is actually dependent on your state, where it was previously thought that brain activity functioned in the same way whether at rest or busy doing a task.
“The second discovery we made was that the blood signal detected by fMRI could reflect the network organisation and cell type distribution,” Associate Professor Chuang said.
“These findings have significant implications for how brain structure shapes function, and how to predict activity based on the knowledge of this structure. More practically, what we now know will impact the design of DBS and other brain stimulation techniques.
“The next steps are to work with clinicians versed in brain stimulation to determine how we can utilise this knowledge combined with human data to help improve our understanding of DBS.”
This more comprehensive understanding could enable us to better predict DBS results and potentially improve its design for better therapeutic outcomes.
About this neurotech and DBS research news
Author: Merrett Pye
Source: University of Queensland
Contact: Merrett Pye – University of Queensland
Image: The image is credited to Associate Professor Kai-Hsiang Chuang / Queensland Brain Institute
Original Research: Closed access.
“Hemodynamic transient and functional connectivity follow structural connectivity and cell type over the brain hierarchy” by Kai-Hsiang Chuang. PNAS
Abstract
Hemodynamic transient and functional connectivity follow structural connectivity and cell type over the brain hierarchy
The neural circuit of the brain is organized as a hierarchy of functional units with wide-ranging connections that support information flow and functional connectivity. Studies using MRI indicate a moderate coupling between structural and functional connectivity at the system level.
However, how do connections of different directions (feedforward and feedback) and regions with different excitatory and inhibitory (E/I) neurons shape the hemodynamic activity and functional connectivity over the hierarchy are unknown.
Here, we used functional MRI to detect optogenetic-evoked and resting-state activities over a somatosensory pathway in the mouse brain in relation to axonal projection and E/I distribution.
Using a highly sensitive ultrafast imaging, we identified extensive activation in regions up to the third order of axonal projections following optogenetic excitation of the ventral posteriomedial nucleus of the thalamus.
The evoked response and functional connectivity correlated with feedforward projections more than feedback projections and weakened with the hierarchy.
The hemodynamic response exhibited regional and hierarchical differences, with slower and more variable responses in high-order areas and bipolar response predominantly in the contralateral cortex.
Electrophysiological recordings suggest that these reflect differences in neural activity rather than neurovascular coupling. Importantly, the positive and negative parts of the hemodynamic response correlated with E/I neuronal densities, respectively. Furthermore, resting-state functional connectivity was more associated with E/I distribution, whereas stimulus-evoked effective connectivity followed structural wiring.
These findings indicate that the structure–function relationship is projection-, cell-type- and hierarchy-dependent. Hemodynamic transients could reflect E/I activity and the increased complexity of hierarchical processing.







