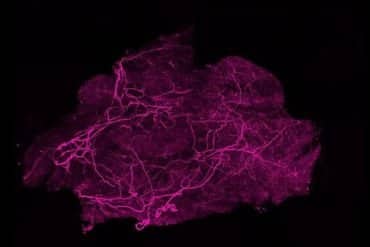
Summary: A new study has discovered that the human brain’s aging process is not unique; mice undergo strikingly similar neural network changes as they grow older. By using specialized fMRI technology to scan awake mice across their lifespans, researchers found that both species experience a breakdown in “modular specialization”—the brain’s ability to maintain distinct, efficient networks for specific tasks.
While humans possess more integrated brain networks (likely fueling higher cognition), they also appear to decline faster than mice. This shared aging trajectory provides a powerful new model for testing anti-aging interventions, such as diet or genetics, in a fraction of the time required for human studies.
Key Facts
- Network Breakdown: Both humans and mice show a decline in how specialized brain modules interact as they age, a hallmark of cognitive decay.
- The “3,000x” Challenge: Because mouse brains are 3,000 times smaller than human brains, researchers used MRI magnets three times stronger than standard clinical scanners to capture fine details.
- Integration vs. Vulnerability: Human brains are more interconnected than mouse brains, which may boost intelligence but also makes them more susceptible to rapid age-related decline.
- Accelerated Research: Because mice age from “18 to 70” in just 17 months, scientists can now test lifelong lifestyle impacts (like diet or medicine) without waiting 80 years for human data.
Source: Zuckerman Institute
By scanning the brains of mice throughout their lifespans, scientists at Columbia’s Zuckerman Institute and the University of Texas at Dallas have discovered that the human brain is not unique in how it changes with age.
These findings might one day help researchers pinpoint mechanisms in humans that confer vulnerability to, or grant resilience from, age-related brain decline, diseases and disorders.
Their research was published online today in Proceedings of the National Academy of Sciences.
The discovery that the brains of humans and mice age in similar ways may one day help scientists understand the factors underlying brain changes during aging. This knowledge can then help researchers find strategies to slow, halt or potentially reverse these declines.
“By looking at mice, we can see if, say, a change in diet in their youth has an effect on them in old age, and we don’t have to wait 80 years for results as we would with humans,” said study co-senior author Itamar Kahn, PhD, a principal investigator at Columbia’s Zuckerman Institute who is also an associate professor of neuroscience at Columbia’s Vagelos College of Physicians and Surgeons.
The human brain, the most complex organ, operates as a network of interconnected modules specialized for tasks, such as perceiving colors or recognizing faces. Previous research found these modules become less specialized as people age, a breakdown associated with worse memory and other forms of cognitive decline.
Much remains unknown about the chemical and cellular machinery underlying this age-related brain decline, as well as ways in which genes, lifestyle, environment or medicine might alter its trajectory. To shed light on this mystery, in the new study, researchers used a non-invasive technology called functional magnetic resonance imaging (fMRI) to scan the brains of 82 mice at several intervals from ages 3 to 20 months, roughly corresponding to ages 18 to 70 years in humans.
fMRI is an imaging method that detects changes in blood flow to the brain. However, the brains of mice are roughly 3,000 times smaller in volume than those of humans, so the researchers needed special strategies for imaging. For instance, the scientists used fMRI scanners with magnetic fields more than three times stronger than ones often used in humans, which let them image smaller details.
Just as most people are awake during MRIs, Dr. Kahn’s lab is one of the few in the world that is able to capture images of the brains of mice while they are awake.
The scientists discovered that aging mice, much like people, experienced a decline in how their different specialized brain modules interacted.
“The way the brain’s modules relate together as a whole is a measure of brain health that appears to apply similarly in both humans and mice,” said study lead author Ezra Winter-Nelson, a doctoral student at the lab of study co-senior author Gagan Wig, PhD, an associate professor of psychology at the University of Texas at Dallas.
The scientists also found significant differences between human and mouse brains. For instance, mouse brain modules communicated less with each other than human ones.
“We think the greater integration that humans have across their brain networks may contribute to aspects of cognition that are especially developed in humans,” Dr. Wig said.
In addition, the human decline in brain module specialization was faster than in mice. “So while we as humans have this ability to integrate information across more widely distributed parts of the brain, that may leave us more vulnerable to brain and cognitive decline when compared to mice,” Dr. Wig added.
The researchers noted that they only investigated one type of lab mice. “We know there are other types of mice that show variability in how they respond to aging,” Dr. Kahn said. “So we want to look at other types of mice to understand how genetics affect trajectories of aging.”
Dr. Kahn says these findings open up ways to study brain aging that would not be possible in humans. Scientists can investigate the effects that genetics, the environment and other factors might have on age-related decline in mice, and with advanced research tools now available.
Previous mouse neuroscience research has drawn criticism for often not possessing clinical relevance in humans. Much of that prior work looked at changes seen at the cellular level.
“What we’re doing is looking at the brain at the network level,” Dr. Kahn noted. “We believe that looking at both the cellular and network level in mice may prove better for developing therapeutic approaches that actually work in humans.”
Funding:
The full list of authors includes Ezra Winter-Nelson, Eyal Bergmann, Micaela Y. Chan, Gabriella Vill, Liang Han, Ziwei Zhang, Alexandra Kavushansky, Irit Dolgopyat, Jad Asleh, Jennifer D. Whitesell, Itamar Kahn and Gagan S. Wig. The authors report no conflicts of interest.
Key Questions Answered:
A: It’s about the “wiring diagram.” While the size differs, the way the brain organizes itself into specialized modules (like “perceiving” vs. “acting”) and then loses that organization with age is remarkably consistent across both species.
A: It’s the price of high performance. Humans have more “integrated” networks, which allows for complex thought, but this high level of connectivity might act like a complex machine—easier to break and faster to degrade when parts start to fail.
A: It significantly speeds up the clock. Instead of waiting decades to see if a supplement or habit helps a human brain stay young, scientists can now get those answers from mice in less than two years using these shared “network” benchmarks.
Editorial Notes:
- This article was edited by a Neuroscience News editor.
- Journal paper reviewed in full.
- Additional context added by our staff.
About this neurology and aging research news
Author: Charles Choi
Source: Zuckerman Institute
Contact: Charles Choi – Zuckerman Institute
Image: The image is credited to Neuroscience News
Original Research: The findings will appear in PNAS