Summary: For years, ketamine has been hailed as a “miracle” fast-acting treatment for depression, but its exact mechanism in the human brain has remained a mystery. Now, a study has used a world-first PET imaging tracer to see exactly what’s happening.
The study reveals that ketamine’s rapid antidepressant effect is driven by the dynamic movement of AMPA receptors—proteins responsible for communication between brain cells. By directly visualizing these receptors in living patients, scientists have proven that ketamine restores “synaptic plasticity,” literally helping the brain’s reward and mood-regulation centers communicate effectively again.
Key Facts
- The AMPAR Mechanism: Ketamine works by modulating the density of AMPA receptors (AMPAR) on the surface of brain cells, which is crucial for brain plasticity and mood regulation.
- The [¹¹C]K-2 Tracer: This breakthrough was made possible by a novel PET tracer that allows scientists to see these specific receptors in a living human brain for the first time.
- Region-Specific Changes: Ketamine doesn’t just “boost” everything; it increases receptor density in the cortex (higher-level thinking) while decreasing it in the habenula (a region linked to reward and disappointment).
- Treatment-Resistant Relief: The study specifically focused on patients who did not respond to traditional antidepressants, confirming ketamine as a powerful tool for the ~30% of patients with TRD.
- Personalized Medicine: AMPAR PET imaging could become a biomarker to predict which patients will respond best to ketamine, moving us toward “precision psychiatry.”
Source: Yokohama City University
Major depressive disorder (MDD) is one of the leading causes of disability worldwide, and approximately 30% of patients develop treatment-resistant depression (TRD), a condition that does not respond adequately to conventional antidepressant therapies.
Although ketamine has emerged as a rapid-acting antidepressant for individuals with TRD, its underlying biological mechanism in the human brain has remained poorly understood, limiting efforts to optimize and personalize treatment.
In a new study published in the journal Molecular Psychiatry on March 05, 2026, a research team led by Professor Takuya Takahashi from the Department of Physiology, Yokohama City University Graduate School of Medicine, Japan, employed an innovative positron emission tomography (PET) imaging approach to directly examine changes in glutamate α-amino-3-hydroxy-5-methyl-4-isoxazole propionic acid receptor (AMPAR), a key protein involved in synaptic plasticity and glutamatergic signaling, in patients receiving ketamine.
Prof. Takahashi noted, “Although ketamine has shown rapid antidepressant effects in patients with treatment-resistant depression, its molecular mechanism in the human brain has remained unclear.”
This advance was made possible by the team’s previously developed PET tracer, [¹¹C]K-2, which enables visualization of cell-surface AMPAR in the living human brain. While preclinical studies have long suggested that ketamine’s antidepressant effects depend on AMPAR activity, this study provides the first direct evidence supporting this mechanism in humans.
The study integrated data from three registered clinical trials conducted in Japan and included 34 patients with TRD and 49 healthy control participants. Patients received intravenous ketamine or a placebo over a two-week period, with PET imaging performed before treatment initiation and after the final infusion.
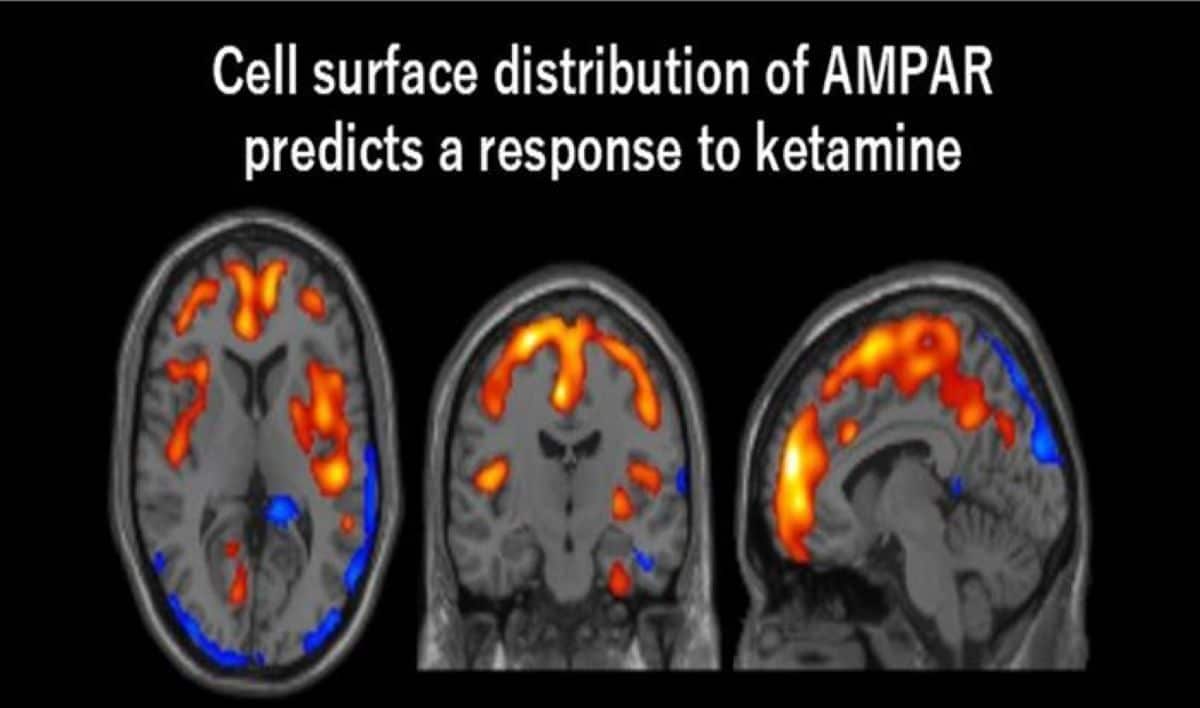
Results revealed that individuals with TRD exhibited widespread, region-specific abnormalities in AMPAR density compared with healthy participants. Notably, ketamine did not induce uniform changes across the brain. Instead, clinical improvement was associated with dynamic, region-specific modulation of AMPAR.
Increases in receptor density were observed in several cortical regions, while decreases were detected in reward-related areas, particularly the habenula. These region-specific changes were strongly correlated with reductions in depressive symptoms.
“Ketamine’s antidepressant effect in patients with TRD is mediated by dynamic changes in AMPAR in the living human brain,” Prof. Takahashi explained.
“Using a novel PET tracer, [¹¹C]K-2, we were able to visualize how ketamine alters AMPAR distribution across specific brain regions and how these changes correlate with improvements in depressive symptoms.”
These findings provide direct human evidence linking molecular mechanisms previously identified in animal models to clinical antidepressant effects.
Beyond advancing mechanistic understanding, the findings have important clinical implications. AMPAR PET imaging may represent a valuable biomarker for evaluating and predicting individual response to ketamine treatment in TRD.
Given the substantial proportion of patients who do not benefit from standard antidepressants, the identification of such biomarkers addresses a critical unmet need in mental healthcare.
By directly visualizing AMPAR dynamics in the living human brain, this study bridges a longstanding gap between preclinical research and clinical psychiatry.
The results establish AMPAR modulation as a central molecular mechanism underlying ketamine’s rapid antidepressant effects and highlight AMPAR PET imaging as a promising tool for guiding personalized treatment strategies.
Ultimately, this work may accelerate the development of more precise, targeted therapies for individuals with treatment-resistant depression.
Key Questions Answered:
A: At controlled, clinical doses, ketamine acts like a “fertilizer” for the brain. Depressed brains often have “withered” synaptic connections. This study shows that ketamine helps the brain physically regrow or relocate these AMPA receptors, allowing neurons to talk to each other again and lifting the symptoms of depression in hours rather than weeks.
A: Until now, we could only guess what ketamine did in humans based on animal studies. The new [¹¹C]K-2 tracer is like a high-definition camera that lets us see the actual molecules moving on the surface of brain cells in a living person. It’s the difference between hearing a description of a house and actually walking through the front door.
A: Yes! Now that we know AMPA receptors are the “engine” of the antidepressant effect, pharmaceutical companies can design new drugs that target these specific receptors directly—potentially without the dissociative side effects of ketamine.
Editorial Notes:
- This article was edited by a Neuroscience News editor.
- Journal paper reviewed in full.
- Additional context added by our staff.
About this ketamine and depression research news
Author: Public Relations Division
Source: Yokohama City University
Contact: Public Relations Division – Yokohama City University
Image: The image is credited to Professor Takuya Takahashi from Yokohama City University Graduate School of Medicine, Japan, and Dr. Hiroyuki Uchida from Keio University School of Medicine, Japan
Original Research: Open access.
“The dynamics of AMPA receptors underlies the efficacy of ketamine in treatment resistant patients with depression” by Waki Nakajima, Mai Hatano, Yohei Ohtani, Hideaki Tani, Taisuke Yatomi, Shohei Tsuchimoto, Yu Fujimoto, Tsuyoshi Eiro, Sadamitsu Ichijo, Kotaro Nakano, Tetsu Arisawa, Yuuki Takada, Kimito Kimura, Hiroki Abe, Akane Sano, Kie Nomoto-Takahashi, Kengo Yonezawa, Sota Tomiyama, Nobuhiro Nagai, Keisuke Kusudo, Shiori Honda, Sotaro Moriyama, Shinichiro Nakajima, Takashige Yamada, Yu Iwabuchi, Masahiro Jinzaki, Kimio Yoshimura, Shariful A. Syed, Sakiko Tsugawa, Hiroyuki Uchida & Takuya Takahashi. Molecular Psychiatry
DOI:10.1038/s41380-026-03510-w
Abstract
The dynamics of AMPA receptors underlies the efficacy of ketamine in treatment resistant patients with depression
Approximately 30% of patients with depression suffer from treatment-resistant depression (TRD). Ketamine has shown antidepressant efficacy for TRD.
While glutamate α-amino-3-hydroxy-5-methyl-4-isoxazolepropionic acid receptor (AMPAR) has been demonstrated to play crucial roles in the process of pharmacological action of ketamine in experimental animals, it remains elusive how ketamine exhibits its efficacy through changes in AMPAR dynamics in patients with TRD.
In this study, using a positron emission tomography (PET) tracer, [11C]K-2, which depicts AMPAR density in the living human brain, we detected a negative correlation between AMPAR density and illness severity and differences in AMPAR distribution between patients with TRD and healthy participants.
Furthermore, we detected brain areas where ketamine administration altered AMPAR density in significant correlations with ketamine-induced antidepressant effect in patients with TRD.
AMPAR density alteration in these regions partially rescued AMPAR phenotype in the affected areas. Thus, AMPAR dynamics underlies the antidepressant effect of ketamine in patients with TRD.