Summary: The activation of dentate granule cells in the hippocampus plays a key role in alterations that occur during the development of post-traumatic epilepsy.
Source: Colorado State University
Traumatic brain injury is a leading cause of epilepsy, a chronic neurological disorder characterized by recurrent seizures that affect around 50 million people.
A research team led by Bret Smith, professor and head of the Department of Biomedical Sciences, discovered specific neuronal processes that could help advance future preventative treatments for post-traumatic epilepsy.
The findings, published in The Journal of Neuroscience, show that activation of a subset of hippocampal neurons plays a key role in the changes that occur during the development of post-traumatic epilepsy and may be restorative.
“We know that trauma induces a cascade of events that can cause epilepsy,” says Smith. “We want to understand exactly what is occurring, and what the endpoints are, and then work backwards to try and stop epilepsy from developing after a brain injury.”
Neuroscience research in Smith’s lab focuses on two distinct programs; one is aimed at identifying neural changes related to the development of epilepsy, which the team has created leading models in the field to study, and the other examines how the brain is influenced by and contributes to hyperglycemia in diabetes.
For this study, Smith’s team looked at neurons called dentate granule cells, which continuously regenerate in areas of the brain that are crucial for learning and memory and are also commonly impacted by epilepsy.
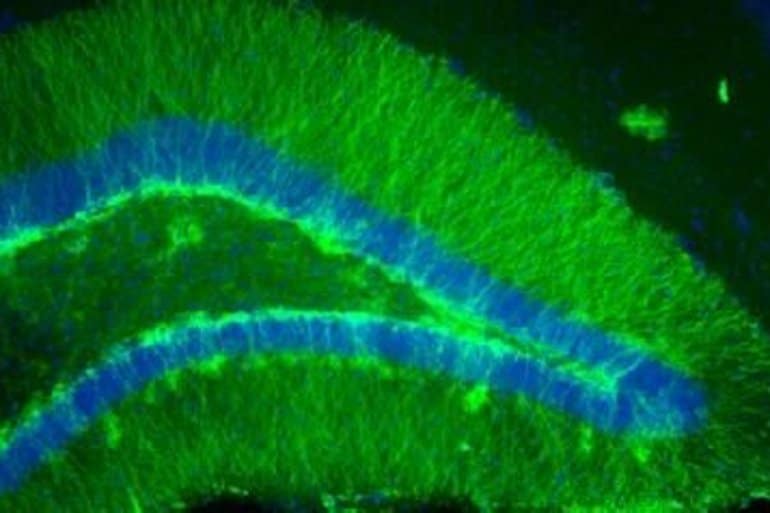
The team was surprised to find that when they were activated, the activity of other brain cells involved in epilepsy were inhibited. And that the cells that were formed just prior to a traumatic brain injury were much more likely to activate this circuit than those generated at other points in time.
“We have been looking at how these synaptic connections reorganize in epilepsy for a long time,” says Smith. “Now, we can look more closely at specific connections of these cells that form at different times.”
Next, Smith’s lab will investigate whether this new connection is restoring function and if it occurs in other types of epilepsy.
“If we can get to a point of understanding the changes that occur in the development of epilepsy well enough, we may be able to prevent or reverse them.”
About this epilepsy research news
Author: Rhea Maze
Source: Colorado State University
Contact: Rhea Maze – Colorado State University
Image: The image is credited to Colorado State University
Original Research: Closed access.
“Adult Born Dentate Granule Cell Mediated Upregulation of Feedback Inhibition in a Mouse Model of Traumatic Brain Injury” by Young-Jin Kang et al. Journal of Neuroscience
Abstract
Adult Born Dentate Granule Cell Mediated Upregulation of Feedback Inhibition in a Mouse Model of Traumatic Brain Injury
Post-traumatic epilepsy (PTE) and behavioral comorbidities frequently develop after traumatic brain injury (TBI). Aberrant neurogenesis of dentate granule cells (DGCs) after TBI may contribute to the synaptic reorganization that occurs in PTE, but how neurogenesis at different times relative to the injury contributes to feedback inhibition and recurrent excitation in the dentate gyrus is unknown.
Thus, we examined whether DGCs born at different postnatal ages differentially participate in feedback inhibition and recurrent excitation in the dentate gyrus using the controlled cortical impact (CCI) model of TBI.
Both sexes of transgenic mice expressing channelrhodopsin2 (ChR2) in postnatally born DGCs were used for optogenetic activation of three DGC cohorts: postnatally early born DGCs, or those born just before or after CCI.
We performed whole-cell patch-clamp recordings from ChR2-negative, mature DGCs and parvalbumin-expressing basket cells (PVBCs) in hippocampal slices to determine whether optogenetic activation of postnatally born DGCs increases feedback inhibition and/or recurrent excitation in mice 8-10 weeks after CCI and whether PVBCs are targets of ChR2-positive DGCs.
In the dentate gyrus ipsilateral to CCI, activation of ChR2-expressing DGCs born before CCI produced increased feedback inhibition in ChR2-negative DGCs and increased excitation in PVBCs compared with those from sham controls.
This upregulated feedback inhibition was less prominent in DGCs born early in life or after CCI. Surprisingly, ChR2-positive DGC activation rarely evoked recurrent excitation in mature DGCs from any cohort.
These results support that DGC birth date-related increased feedback inhibition in of DGCs may contribute to altered excitability after TBI.






