Implications for personalized medicine and molecularly targeted therapies.
Researchers at University of California San Diego School of Medicine have demonstrated that distinct types of glioblastoma, the most common form of brain cancer in adults, tend to develop in different regions of the brain. This finding provides an explanation for how the same cancer-causing mutation can give rise to different types of brain malignancies. Results of the study were published in the May 2, 2016 online edition of Oncotarget.
“It is now well-documented that cancers that look the same under the microscope actually contain different genetic changes, or mutations, and respond differently to therapy,” said Clark Chen, MD, PhD, senior author and vice-chair of research and academic development in the Division of Neurosurgery at UC San Diego School of Medicine. “What remains unclear is how the exact, same mutation can give rise to different subtypes of tumor.”
To study this mystery, Chen’s team developed a new computational method to define where different glioblastoma subtypes develop in the brain. Using clinical images derived from 217 brain tumor patients, Chen’s team discovered that proneural and neural glioblastoma subtypes tend to occur closer to the center of the brain in a region called the subventricular zone (SVZ). In contrast, mesenchymal and classical glioblastoma subtypes tend to develop farther from this region.
The subventricular zone is a unique region of the brain where neural stem cells, which ultimately give rise to all cell types in the brain, reside. During brain development, these stem cells migrate outward from the center region. During this process, the stem cells transform into the dozen or so different cell types that compose the human brain.
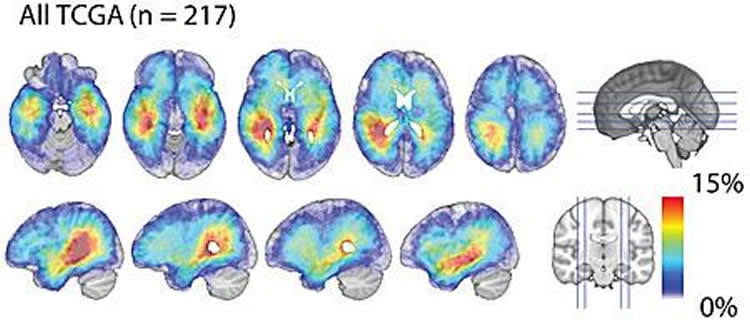
“Our study suggests that if a cancer-causing mutation occurs in the neural stem cell population in the SVZ, it gives rise to the proneural or the neural glioblastoma subtype. On the other hand, if the same mutation occurs in a different cell population located farther away from the SVZ, it will give rise to other subtypes,” said Chen.
Through a mouse model of glioblastoma developed by Lionel Chow, MD, PhD, a pediatric hematologist-oncologist at Cincinnati Children’s Hospital, Chen’s team was able to confirm this hypothesis. In Chow’s model, all glioblastomas arise as a result of the same mutations. Nevertheless, the brain tumors that form closer to the SVZ tend to be proneural or neural subtypes, while tumors form farther from the SVZ tend to be classical or mesenchymal subtypes.
“Because glioblastoma subtypes respond differently to distinct therapies, subtype discrimination will be increasingly important,” said Bob Carter, MD, PhD, chair of neurosurgery at UC San Diego Health. “Refinement of this non-invasive method for determining glioblastoma subtypes may achieve the goal of personalizing glioblastoma therapy without subjecting our patients to surgery.”
Funding: Support for this study came, in part, from the Doris Duke Charitable Foundation, Sontag Foundation, Burroughs Wellcome Fund, Forbeck Foundation and Kimmel Foundation.
Contributors to this paper include: Tyler C. Steed, Jeffrey M. Treiber, Kunal Patel, Valya Ramakrishnan, Bob S. Carter, and Anders M. Dale, UC San Diego; and Lionel M. L. Chow, Alexander Merk and Amanda R. Smith, Cincinnati Children’s Hospital Medical Center.
Source: Jackie Carr – UCSD
Image Source: The image is credited to UC San Diego Health.
Original Research: Abstract for “Differential localization of glioblastoma subtype: implications on glioblastoma pathogenesis” by Tyler C. Steed, Jeffrey M. Treiber, Kunal Patel, Valya Ramakrishnan, Alexander Merk, Amanda R. Smith, Bob S. Carter, Anders M. Dale, Lionel M. L. Chow, and Clark C. Chen in Oncotarget. Published online May 2 2016 doi:10.18632/oncotarget.8551
Abstract
Differential localization of glioblastoma subtype: implications on glioblastoma pathogenesis
Introduction: The subventricular zone (SVZ) has been implicated in the pathogenesis of glioblastoma. Whether molecular subtypes of glioblastoma arise from unique niches of the brain relative to the SVZ remains largely unknown. Here, we tested whether these subtypes of glioblastoma occupy distinct regions of the cerebrum and examined glioblastoma localization in relation to the SVZ.
Methods: Pre-operative MR images from 217 glioblastoma patients from The Cancer Imaging Archive were segmented automatically into contrast enhancing (CE) tumor volumes using Iterative Probabilistic Voxel Labeling (IPVL). Probabilistic maps of tumor location were generated for each subtype and distances were calculated from the centroid of CE tumor volumes to the SVZ. Glioblastomas that arose in a Genetically Modified Murine Model (GEMM) model were also analyzed with regard to SVZ distance and molecular subtype.
Results: Classical and mesenchymal glioblastomas were more diffusely distributed and located farther from the SVZ. In contrast, proneural and neural glioblastomas were more likely to be located in closer proximity to the SVZ. Moreover, in a GFAP-CreER; PtenloxP/loxP; Trp53loxP/loxP; Rb1loxP/loxP; Rbl1-/- GEMM model of glioblastoma where tumor can spontaneously arise in different regions of the cerebrum, tumors that arose near the SVZ were more likely to be of proneural subtype (p < 0.0001).
Conclusions: Glioblastoma subtypes occupy different regions of the brain and vary in proximity to the SVZ. These findings harbor implications pertaining to the pathogenesis of glioblastoma subtypes.
“Differential localization of glioblastoma subtype: implications on glioblastoma pathogenesis” by Tyler C. Steed, Jeffrey M. Treiber, Kunal Patel, Valya Ramakrishnan, Alexander Merk, Amanda R. Smith, Bob S. Carter, Anders M. Dale, Lionel M. L. Chow, and Clark C. Chen in Oncotarget. Published online May 2 2016 doi:10.18632/oncotarget.8551






