Some responses come automatically, like reflexes. Others vary with circumstance and experience. A once-delicious smell can be easily overlooked during a stressful moment or when it calls to mind about of food poisoning, for instance. This happens because, within the brain, molecules known as neuromodulators reroute information about that odor.
New research at The Rockefeller University takes advantage of the simple architecture within the fruit fly brain to examine how one such molecule, dopamine, acts like an operator at a switchboard. The results, published Dec. 17, 2015 in Cell, show how it changes the flow of information, the basis for flexibility in behavior.
“When we took a close look at a region of the fly brain involved in processing odor, we saw that the release of dopamine within it can alter the strength of connections between neurons with exquisite precision,” says senior author Vanessa Ruta, Gabrielle H. Reem and Herbert J. Kayden Assistant Professor and head of the Laboratory of Neurophysiology and Behavior. “This has the effect of altering the messages relayed to other parts of the brain, which could in turn lead to different responses to identical odors.”
The region, known as the mushroom body because of its shape, is responsible for a fly’s ability to learn to associate an odor with a reward or a punishment. Ruta, first author Raphael Cohn, a graduate student in the lab, and co-author Ianessa Morrante, were also interested in how the animal’s internal state — whether it was active or quiet, for instance — might influence dopamine, and as a result, information processing.
The simple wiring of the fly’s mushroom body made it an ideal subject; a similar study would have been very challenging in the more complex mammalian brain. Signals from the fly’s nose, its antennae, travel into the mushroom body via the branches of neurons known as Kenyon cells. These branches traverse distinct compartments, and within each compartment they connect both with dopamine-producing neurons and with output neurons, which signal to other parts of the brain.
While the human brain does not have a mushroom body, it does have dopamine. This neurotransmitter is present throughout the animal kingdom, and is known to play a number of important roles in human brain function, including in learning and motivation, where it functions as a neuromodulator.
In their experiments, Ruta and her collaborators found that dopamine neurons reaching into two of four compartments became active when the fly was given a tasty sugar solution, while the other two became active when it experienced a briefly painful shock. The same reciprocal pattern of activity showed up while the fly was attached to the microscope slide, either motionless or flailing its legs wildly, with no outside stimulus.
The latter result confirmed the dopamine signaling isn’t just involved in learning; it is linked to a fly’s internal state. “It appears these pathways create a beautiful moment-by-moment representation of the animal’s ongoing experience, generating much more complex and rich patterns than was previously appreciated,” Ruta says.
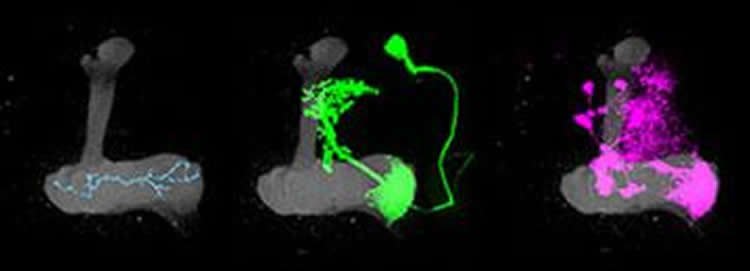
To better understand the interactions among neurons in the mushroom body, they engineered Kenyon cells so their terminal ends at synapses glowed as a result of signaling. This revealed that the same Kenyon cell may or may not send a signal to the output neurons depending on the compartment in which the two come together. They also found that the dopamine neurons determine whether that signal is sent.
“When we artificially activated the dopamine neurons or provided external stimuli, such as a sugar reward, we could change the output neurons’ responses to odors,” Cohn says. “Essentially, the dopamine signaling is rerouting the course of the information along the branch of the Kenyon cell and determining to which output neuron it goes.”
This discovery quite likely parallels the action of dopamine in the human brain, the researchers say.
“In the mammalian brain, dopamine neurons have more and more complicated connections to many brain centers,” Ruta says. “Even so, human dopamine neurons probably work in a coordinated manner to create the basis for flexible behavior, as we saw in the fly brain.”
Source: Katherine Fenz – Rockefeller University
Image Credit: The image is credited to Laboratory of Neurophysiology and Behavior at The Rockefeller University/Neuron
Original Research: Abstract for “Coordinated and Compartmentalized Neuromodulation Shapes Sensory Processing in Drosophila” by Raphael Cohn, Ianessa Morantte, and Vanessa Ruta in Cell. Published online December 16 2015 doi:10.1016/j.cell.2015.11.019
Abstract
Coordinated and Compartmentalized Neuromodulation Shapes Sensory Processing in Drosophila
Highlights
•Mushroom body dopaminergic neurons act in concert to represent contextual cues
•Dopamine bidirectionally modifies synapses in precise domains along Kenyon cell axons
•Odor signals are differentially conveyed to each postsynaptic target of a Kenyon cell
•Activity of output pathways depends on an animal’s external context or internal state
Summary
Learned and adaptive behaviors rely on neural circuits that flexibly couple the same sensory input to alternative output pathways. Here, we show that the Drosophila mushroom body functions like a switchboard in which neuromodulation reroutes the same odor signal to different behavioral circuits, depending on the state and experience of the fly. Using functional synaptic imaging and electrophysiology, we reveal that dopaminergic inputs to the mushroom body modulate synaptic transmission with exquisite spatial specificity, allowing individual neurons to differentially convey olfactory signals to each of their postsynaptic targets. Moreover, we show that the dopaminergic neurons function as an interconnected network, encoding information about both an animal’s external context and internal state to coordinate synaptic plasticity throughout the mushroom body. Our data suggest a general circuit mechanism for behavioral flexibility in which neuromodulatory networks act with synaptic precision to transform a single sensory input into different patterns of output activity.
“Coordinated and Compartmentalized Neuromodulation Shapes Sensory Processing in Drosophila” by Raphael Cohn, Ianessa Morantte, and Vanessa Ruta in Cell. Published online December 16 2015 doi:10.1016/j.cell.2015.11.019






