Summary: Newly discovered sensory neurons send information related to stress and metabolism from adipose fat tissue to the brain.
Source: Scripps Research Institute
What did the fat say to the brain? For years, it was assumed that hormones passively floating through the blood were the way that a person’s fat—called adipose tissue—could send information related to stress and metabolism to the brain.
Now, Scripps Research scientists report in Nature that newly identified sensory neurons carry a stream of messages from adipose tissue to the brain.
“The discovery of these neurons suggests for the first time that your brain is actively surveying your fat, rather than just passively receiving messages about it,” says co-senior author Li Ye, PhD, the Abide-Vividion Chair in Chemistry and Chemical Biology and an associate professor of neuroscience at Scripps Research. “The implications of this finding are profound.”
“This is yet another example of how important sensory neurons are to health and disease in the human body,” says co-senior author and professor Ardem Patapoutian, PhD, who is also a Nobel laureate and a Howard Hughes Medical Institute investigator.
In mammals, adipose tissue stores energy in the form of fat cells and, when the body needs energy, releases those stores. It also controls a host of hormones and signaling molecules related to hunger and metabolism. In diseases including diabetes, fatty liver disease, atherosclerosis and obesity, that energy storage and signaling often goes awry.
Researchers have long known that nerves extend into adipose tissue, but suspected they weren’t sensory neurons that carry data to the brain. Instead, most hypothesized that the nerves in fat belonged mostly to the sympathetic nervous system—the network responsible for our fight-or-flight response, which switches on fat-burning pathways during times of stress and physical activity.
Attempts to clarify the types and functions of these neurons have been difficult; methods used to study neurons closer to the surface of the body or in the brain don’t work well deep in adipose tissue, where nerves are hard to see or to stimulate.
Ye and colleagues developed two new methods that enable them to overcome these challenges. First, an imaging approach called HYBRiD turned mouse tissues transparent and allowed the team to better track the paths of neurons as they snaked into adipose tissue.
The researchers discovered that nearly half of these neurons didn’t connect to the sympathetic nervous system, but instead to dorsal root ganglia—an area of the brain where all sensory neurons originate.
To better probe the role of these neurons in adipose tissue, the group turned to a second new technique, which they named ROOT, for “retrograde vector optimized for organ tracing”. ROOT let them selectively destroy small subsets of sensory neurons in the adipose tissue using a targeted virus and then observe what happened.
“This research was really made possible by the way these new methods came together,” says Yu Wang, a graduate student in both the Ye and Patapoutian labs and first author of the new paper. “When we first started this project, there weren’t existing tools to answer these questions.”
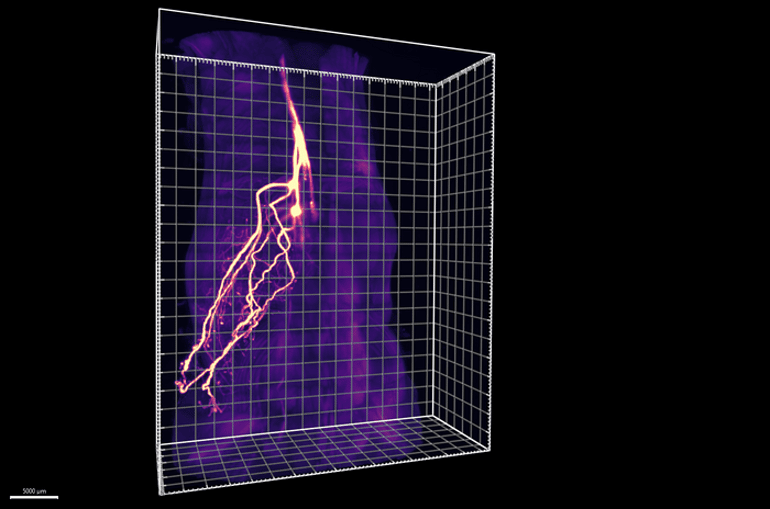
The experiments revealed that when the brain doesn’t receive sensory messages from adipose tissue, programs triggered by the sympathetic nervous system—related to the conversion of white fat to brown fat—become overly active in fat cells, resulting in a larger than normal fat pad with especially high levels of brown fat, which breaks down other fat and sugar molecules to produce heat. Indeed, the animals with blocked sensory neurons—and high levels of sympathetic signaling—had increased body temperatures.
The findings suggest that the sensory neurons and sympathetic neurons might have two opposing functions, with sympathetic neurons needed to turn on fat burning and the production of brown fat, and sensory neurons required to turn these programs down.
“This tells us that there’s not just a one-size-fits-all instruction that brain sends adipose tissue,” says Li. “It’s more nuanced than that; these two types of neurons are acting like a gas pedal and a brake for burning fat.”
The team doesn’t yet know exactly what messages the sensory neurons convey to the brain from adipose tissue, only that the connections and communications are key for keeping fat healthy. They are planning future research into what the neurons are sensing and whether other similar cells exist in additional internal organs.
Funding: Other authors of the paper “The role of somatosensory innervation of adipose tissue,” were Yu Wang, Verina Leung, Yunxiao Zhang, Victoria S. Nudell, Meaghan Loud, M. Rocio Servin-Vences, Dong Yang and Kristina Wang of Scripps Research; and Maria Dolores Moya-Garzon, Veronica L. Li, and Jonathan Z. Long of Stanford University.
Funding was provided by the Howard Hughes Medical Institute, the National Institutes of Health (R35 NS105067, NIH Director’s New Innovator Award DP2DK128800, NIDDK K01DK114165), the Whitehall Foundation, the Baxter Foundation, a Helen Dorris Scholars fellowship, a Damon Runyon Cancer Research Foundation Merck fellowship (DRG-2405-20), and a Fundacion Alfonso Martin Escudero postdoctoral fellowship.
About this neuroscience research news
Author: Press Office
Source: Scripps Research Institute
Contact: Press Office – Scripps Research Institute
Image: The image is credited to Scripps Research Institute
Original Research: Open access.
“The role of somatosensory innervation of adipose tissue” by Li Ye et al. Nature
Abstract
The role of somatosensory innervation of adipose tissue
Adipose tissues communicate with the central nervous system to maintain whole-body energy homeostasis.
The mainstream view is that circulating hormones secreted by the fat convey the metabolic state to the brain, which integrates peripheral information and regulates adipocyte function through noradrenergic sympathetic output. Moreover, somatosensory neurons of the dorsal root ganglia innervate adipose tissue.
However, the lack of genetic tools to selectively target these neurons has limited understanding of their physiological importance.
Here we developed viral, genetic and imaging strategies to manipulate sensory nerves in an organ-specific manner in mice. This enabled us to visualize the entire axonal projection of dorsal root ganglia from the soma to subcutaneous adipocytes, establishing the anatomical underpinnings of adipose sensory innervation.
Functionally, selective sensory ablation in adipose tissue enhanced the lipogenic and thermogenetic transcriptional programs, resulting in an enlarged fat pad, enrichment of beige adipocytes and elevated body temperature under thermoneutral conditions.
The sensory-ablation-induced phenotypes required intact sympathetic function. We postulate that beige-fat-innervating sensory neurons modulate adipocyte function by acting as a brake on the sympathetic system.
These results reveal an important role of the innervation by dorsal root ganglia of adipose tissues, and could enable future studies to examine the role of sensory innervation of disparate interoceptive systems.






