Summary: In lower mammals, the iris may sense light and cause the pupils to constrict without involving the brain, a new study reports.
Source: Johns Hopkins Medicine.
Experimenting with mice, neuroscientists at Johns Hopkins Medicine report new evidence that the eye’s iris in many lower mammals directly senses light and causes the pupil to constrict without involving the brain.
In a report in the June 19 issue of the journal Current Biology, the researchers detail how the pupils in a mouse’s eyes get smaller when the animal is moved from a dark to a lit room even when the nerve connections between the animal’s brain and eyes are severed.
Their findings prove that mouse eyes have a photosensitive function built directly into the ring of sphincter muscle surrounding the pupil.
The mammalian eye adapts to changing light conditions by constricting or enlarging the pupil. That action, referred to as pupillary light reflex, is controlled by opposing dilator and sphincter muscles in the iris. “The traditional view of this reflex is that light triggers nerve signals traveling from the eye’s retina to the brain, thereby activating returning nerve signals, relayed by the neurotransmitter acetylcholine, that make the sphincter muscle contract and constrict the pupil,” says King-Wai Yau, Ph.D., a neuroscientist at the Johns Hopkins University School of Medicine and an author of the report.
But research by Yau and his team several years ago, building on earlier work by others, showed that nocturnal mammals and mammals active at dawn and dusk, such as mice, rabbits, cats and dogs, don’t need the brain for this reflex to work. Instead, Yau’s lab found that even when isolated, the sphincter muscle contracts in response to light, employing a light-sensitive pigment called melanopsin.
The simplest explanation for that observation would be that the muscle is itself sensitive to light. However, other researchers offered the alternative explanation that there might be light-sensitive nerve fibers containing melanopsin present on the sphincter muscle that make the muscle contract by piggybacking on the brain’s pupillary light reflex circuitry involving acetylcholine.
This suggestion prompted Yau’s team to ask what would happen if the action of acetylcholine were blocked. “Sure enough, even after blocking the action of acetylcholine pharmacologically, the isolated iris sphincter muscle still contracted in response to light, adding confidence to the notion that the muscle is itself light-sensitive because it contains melanopsin,” Yau says of the present work.
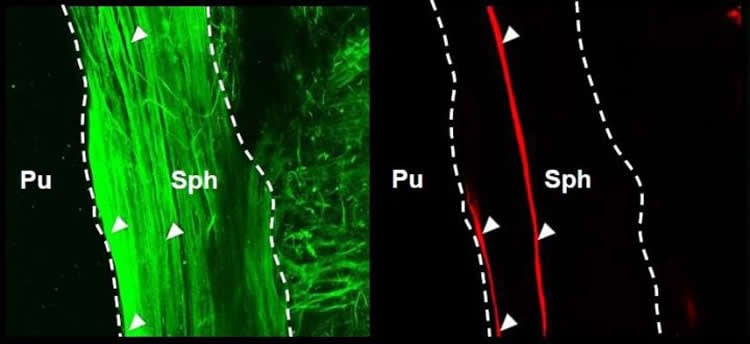
To go further, they used genetics to selectively prevent muscle from making melanopsin. Indeed, this approach removed the effect of light on the sphincter muscle, even when acetylcholine’s action was intact. “We thus have convincingly proven that the sphincter muscle is intrinsically light-sensitive, a very unusual property for muscle,” Yau says.
It turns out lower mammals that are active during the day, such as ground squirrels, lack the local pupillary light reflex Yau’s team found; the same applies to primates, whether nocturnal or not. “The broad picture,” Yau says, “is that the local pupillary light reflex appeared early in primitive vertebrates such as jawless fish, even before the brain got involved. In the course of evolution, the brain became involved in this reflex, but the local mechanism remained dominant in amphibians. By the time mammals appeared, the local reflex was progressively less important, becoming extinct altogether in subprimates that are active during the day and in primates. It’s the local light reflex’s absence in human beings that allows doctors to quickly evaluate whether a comatose patient is brain-dead by checking his or her pupillary light reflex.”
Qian Wang, a graduate student under Yau, carried out the majority of the experiments. Other researchers involved include Wendy Wing Sze Yue, Zheng Jiang, and Dwight E. Bergles, all of Johns Hopkins; Shin H. Kang of Temple University; Tian Xue of the University of Science and Technology of China; Katsuhiko Mikoshiba of the RIKEN Brain Science Institute in Japan; and Stefan Offermanns of the Max Planck Institute for Heart and Lung Research in Germany.
Funding: This work was funded by the National Eye Institute (grant number EY14596); the António Champalimaud Vision Award, Portugal; the National Institute of Neurological Disorders and Stroke (grant number P30 NS050274); and a Howard Hughes Medical Institute International Predoctoral Fellowship to Yue.
Source: Rachel Butch – Johns Hopkins Medicine
Image Source: NeuroscienceNews.com image is credited to Johns Hopkins Medicine.
Original Research: Abstract for “Synergistic Signaling by Light and Acetylcholine in Mouse Iris Sphincter Muscle” by Qian Wang, Wendy Wing Sze Yue, Zheng Jiang, Tian Xue, Shin H. Kang10, Dwight E. Bergles, Katsuhiko Mikoshiba, Stefan Offermanns, and King-Wai Yau in Current Biology. Published online June 1 2017 doi:10.1016/j.cub.2017.05.022
[cbtabs][cbtab title=”MLA”]Johns Hopkins Medicine “A No-Brainer? Mouse Eyes Constrict to Light Without Direct Link to the Brain.” NeuroscienceNews. NeuroscienceNews, 19 June 2017.
<https://neurosciencenews.com/eye-constriction-brain-6929/>.[/cbtab][cbtab title=”APA”]Johns Hopkins Medicine (2017, June 19). A No-Brainer? Mouse Eyes Constrict to Light Without Direct Link to the Brain. NeuroscienceNew. Retrieved June 19, 2017 from https://neurosciencenews.com/eye-constriction-brain-6929/[/cbtab][cbtab title=”Chicago”]Johns Hopkins Medicine “A No-Brainer? Mouse Eyes Constrict to Light Without Direct Link to the Brain.” https://neurosciencenews.com/eye-constriction-brain-6929/ (accessed June 19, 2017).[/cbtab][/cbtabs]
Abstract
Synergistic Signaling by Light and Acetylcholine in Mouse Iris Sphincter Muscle
Highlights
•The pupillary light reflex of many sub-primates has a mechanism intrinsic to the iris
•The mouse iris sphincter muscle is photosensitive, independent of synaptic transmission
•Deleting melanopsin in sphincter muscle removes its intrinsic photosensitivity
•The components of the melanopsin photosignaling in sphincter muscle are revealed
Summary
The mammalian pupillary light reflex (PLR) involves a bilateral brain circuit whereby afferent light signals in the optic nerve ultimately drive iris-sphincter-muscle contraction via excitatory cholinergic parasympathetic innervation. Additionally, the PLR in nocturnal and crepuscular sub-primate mammals has a “local” component in the isolated sphincter muscle, as in amphibians, fish, and bird. In mouse, this local PLR requires the pigment melanopsin, originally found in intrinsically photosensitive retinal ganglion cells (ipRGCs). However, melanopsin’s presence and effector pathway locally in the iris remain uncertain. The sphincter muscle itself may express melanopsin, or its cholinergic parasympathetic innervation may be modulated by suggested intraocular axonal collaterals of ipRGCs traveling to the eye’s ciliary body or even to the iris. Here, we show that the muscarinic receptor antagonist, atropine, eliminated the effect of acetylcholine (ACh), but not of light, on isolated mouse sphincter muscle. Conversely, selective genetic deletion of melanopsin in smooth muscle mostly removed the light-induced, but not the ACh-triggered, increase in isolated sphincter muscle’s tension and largely suppressed the local PLR in vivo. Thus, sphincter muscle cells are bona fide, albeit unconventional, photoreceptors. We found melanopsin expression in a small subset of mouse iris sphincter muscle cells, with the light-induced contractile signal apparently spreading through gap junctions into neighboring muscle cells. Light and ACh share a common signaling pathway in sphincter muscle. In summary, our experiments have provided details of a photosignaling process in the eye occurring entirely outside the retina.
“Synergistic Signaling by Light and Acetylcholine in Mouse Iris Sphincter Muscle” by Qian Wang, Wendy Wing Sze Yue, Zheng Jiang, Tian Xue, Shin H. Kang10, Dwight E. Bergles, Katsuhiko Mikoshiba, Stefan Offermanns, and King-Wai Yau in Current Biology. Published online June 1 2017 doi:10.1016/j.cub.2017.05.022






