Summary: Stimulating the posterior cingulate cortex increased hippocampal gamma-band power and strengthened the connections between the two brain regions. Using deep brain stimulation could help to treat memory disorders and cognitive impairment.
Source: SfN
Deep brain stimulation of the cingulate cortex worsens memory recall, according to research in epilepsy patients published in Journal of Neuroscience. The technique could be a way to target specific brain areas in the treatment of memory disorders.
Vaidehi Natu and colleagues at the University of Texas Southwestern Medical Center stimulated the posterior cingulate cortex, a brain region involved in memory, using electrodes inserted into the brains of epilepsy patients to treat seizures. The participants were shown a list of words, distracted, and then asked to recall the words.
Natu’s team discovered that stimulating the cingulate cortex reduced the number of recalled words and modified hippocampal brain waves compared to no stimulation. This establishes a direct role for the cingulate cortex in memory encoding and demonstrates that memories can be manipulated via deep brain stimulation, which has implications for the treatment of memory disorders.
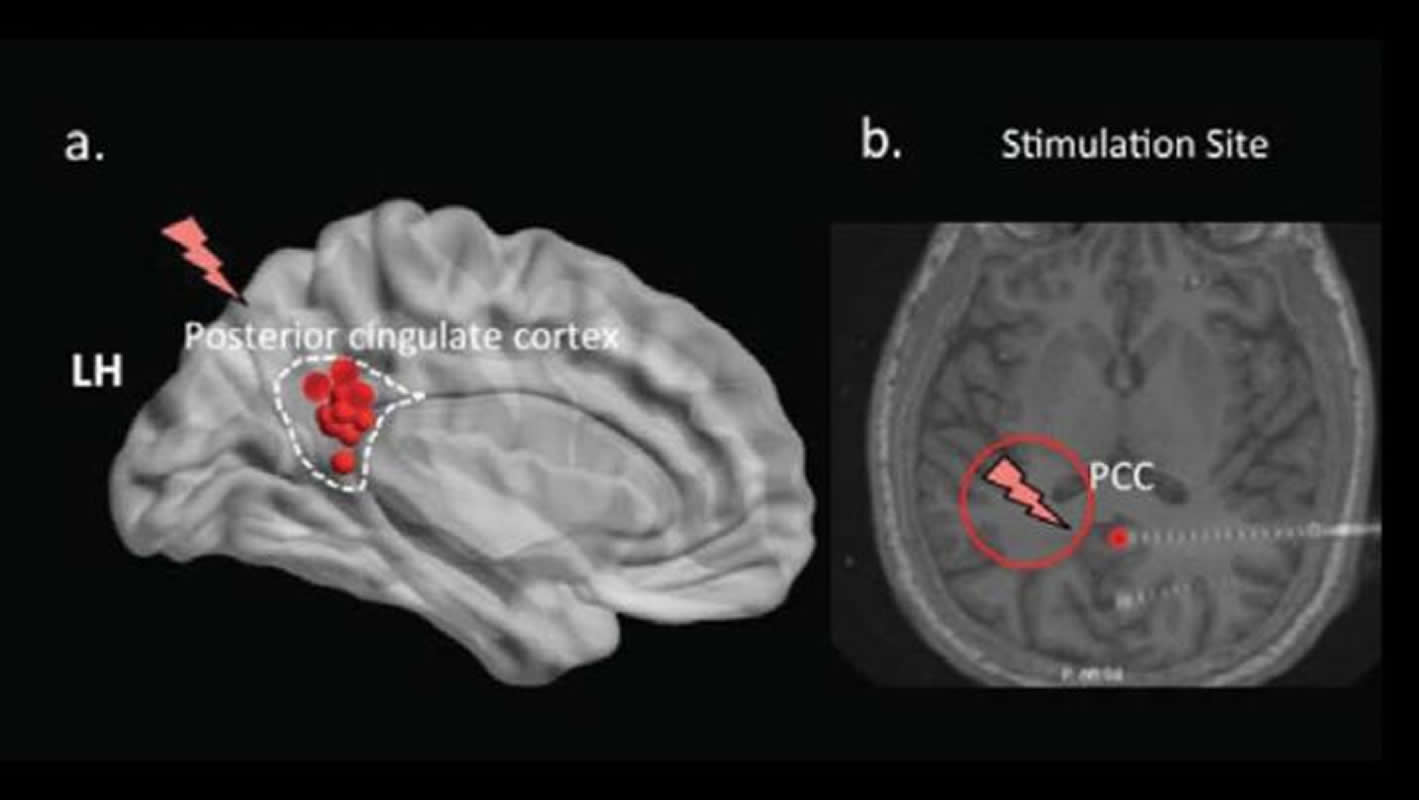
Source:
SfN
Media Contacts:
Calli McMurray – SfN
Image Source:
The image is credited to Natu et al., JNeurosci 2019.
Original Research: Open access
“Stimulation of the posterior cingulate cortex impairs episodic memory encoding”. Vaidehi S. Natu, Jui-Jui Lin, Alexis Burks, Akshay Arora, Michael D. Rugg and Bradley Lega.
Journal of Neuroscience. doi:10.1523/JNEUROSCI.0698-19.2019
Abstract
Stimulation of the posterior cingulate cortex impairs episodic memory encoding
Neuroimaging experiments implicate the posterior cingulate cortex (PCC) in episodic memory processing, making it a potential target for responsive neuromodulation strategies outside of the hippocampal network. However, causal evidence for the role PCC plays in memory encoding is lacking. In human female and male participants (N=17) undergoing seizure mapping, we investigated functional properties of the PCC using deep brain stimulation (DBS) and stereotactic electroencephalography (stereo EEG). We used a verbal free recall paradigm in which the PCC was stimulated during presentation of half of the study lists, while no stimulation was applied during presentation of the remaining lists. We examined whether stimulation affected memory and modulated hippocampal activity. Results revealed four main findings. (i) Stimulation during episodic memory encoding impaired subsequent free recall, predominantly for items presented early in the study lists. (ii) PCC stimulation increased hippocampal gamma band power. (iii) Stimulation-induced hippocampal gamma power predicted the magnitude of memory impairment. (iv) Functional connectivity between the hippocampus and PCC predicted the strength of the stimulation effect on memory. Our findings offer causal evidence implicating the PCC in episodic memory encoding. Importantly, the results indicate that stimulation targeted outside of the temporal lobe can modulate hippocampal activity and impact behavior. Furthermore, measures of connectivity between brain regions within a functional network can be informative in predicting behavioral effects of stimulation. Our findings have significant implications for developing therapies to treat memory disorders and cognitive impairment using DBS.
SIGNIFICANCE STATEMENT
Cognitive impairment and memory loss are critical public health challenges. Deep brain stimulation (DBS) is a promising tool for developing strategies to ameliorate memory disorders by targeting brain regions involved in mnemonic processing. Using DBS, our study sheds light on the lesser-known role of the posterior cingulate cortex (PCC) in memory encoding. Stimulating the PCC during encoding impairs subsequent recall memory. The degree of impairment is predicted by stimulation-induced hippocampal gamma oscillations and functional connectivity between PCC and hippocampus. Our findings provide the first causal evidence implicating PCC in memory encoding and highlight the PCC as a favorable target for neuromodulation strategies, using a-priori connectivity measures to predict stimulation effects. This has significant implications for developing therapies for memory diseases.






