Summary: SARS_CoV_2, the virus responsible for COVID-19 infects and replicates in astrocytes, reducing neural viability.
Source: FAPESP
A Brazilian study published in the journal PNAS describes some of the effects infection by SARS-CoV-2 can have on the central nervous system.
A preliminary version (not yet peer-reviewed) posted in 2020 was one of the first to show that the virus that causes COVID-19 can infect brain cells, especially astrocytes. It also broke new ground by describing alterations in the structure of the cortex, the most neuron-rich brain region, even in cases of mild COVID-19.
The cerebral cortex is the outer layer of gray matter over the hemispheres. It is the largest site of neural integration in the central nervous system and plays a key role in complex functions such as memory, attention, consciousness, and language.
The investigation was conducted by several groups at the State University of Campinas (UNICAMP) and the University of São Paulo (USP). Researchers at the Brazilian Biosciences National Laboratory (LNBio), D’Or Institute (IDOR) and the Federal University of Rio de Janeiro (UFRJ) also contributed to the study.
“Two previous studies detected the presence of the novel coronavirus in the brain, but no one knew for sure if it was in the bloodstream, endothelial cells [lining the blood vessels] or nerve cells. We showed for the first time that it does indeed infect and replicate in astrocytes, and that this can reduce neuron viability,” Daniel Martins-de-Souza, one of the leaders of the study, told Agência FAPESP. Martins-de-Souza is a professor at UNICAMP’s Biology Institute and a researcher affiliated with IDOR.
Astrocytes are the most abundant central nervous system cells. Their functions include providing biochemical support and nutrients for neurons; regulating levels of neurotransmitters and other substances that may interfere with neuronal functioning, such as potassium; maintaining the blood-brain barrier that protects the brain from pathogens and toxins; and helping to maintain brain homeostasis.
Infection of astrocytes was confirmed by experiments using brain tissue from 26 patients who died of COVID-19. The tissue samples were collected during autopsies conducted using minimally invasive procedures by Alexandre Fabro, a pathologist and professor at the University of São Paulo’s Ribeirão Preto Medical School (FMRP-USP). The analysis was coordinated by Thiago Cunha, also a professor in FMRP-USP and a member of the Center for Research on Inflammatory Diseases (CRID).
The researchers used a technique known as immunohistochemistry, a staining process in which antibodies act as markers of viral antigens or other components of the tissue analyzed.
“For example, we can insert one antibody into the sample to turn the astrocytes red on binding to them, another to mark the SARS-CoV-2 spike protein by making it green, and a third to highlight the virus’s double-stranded RNA, which only appears during replication, by turning it magenta,” Martins-de-Souza explained.
“When the images produced during the experiment were overlaid, all three colors appeared simultaneously only in astrocytes.”
According to Cunha, the presence of the virus was confirmed in five of the 26 samples analyzed. Alterations suggesting possible damage to the central nervous system were also found in these five samples.
“We observed signs of necrosis and inflammation, such as edema [swelling caused by a buildup of fluid], neuronal lesions and inflammatory cell infiltrates,” he said.
The capacity of SARS-CoV-2 to infect brain tissue and its preference for astrocytes were confirmed by Adriano Sebolella and his group at FMRP-USP using the method of brain-derived slice cultures, an experimental model in which human brain tissue obtained during surgery to treat neurological diseases such as drug-refractory epilepsy, for example, is cultured in vitro and infected with the virus.
Persistent symptoms
In another part of the research, conducted in UNICAMP’s School of Medical Sciences (FCM), 81 volunteers who had recovered from mild COVID-19 were submitted to magnetic resonance imaging (MRI) scans of their brains.
These scans were performed 60 days after diagnostic testing on average. A third of the participants still had neurological or neuropsychiatric symptoms at the time. They complained mostly of headache (40%), fatigue (40%), memory alterations (30%), anxiety (28%), loss of smell (28%), depression (20%), daytime drowsiness (25%), loss of taste (16%) and low libido (14%).
“We posted a link for people interested in participating in the trial to register, and were surprised to get more than 200 volunteers in only a few days. Many were polysymptomatic, with widely varying complaints. In addition to the neuroimaging exam, they’re being evaluated neurologically and taking standardized tests to measure performance in cognitive functions such as memory, attention and mental flexibility. In the article we present the initial results,” said Clarissa Yasuda, a professor and member of the Brazilian Research Institute for Neuroscience and Neurotechnology (BRAINN).
Only volunteers diagnosed with COVID-19 by RT-PCR and not hospitalized were included in the study. The assessments were carried out after the end of the acute phase, and the results were compared with data for 145 healthy uninfected subjects.
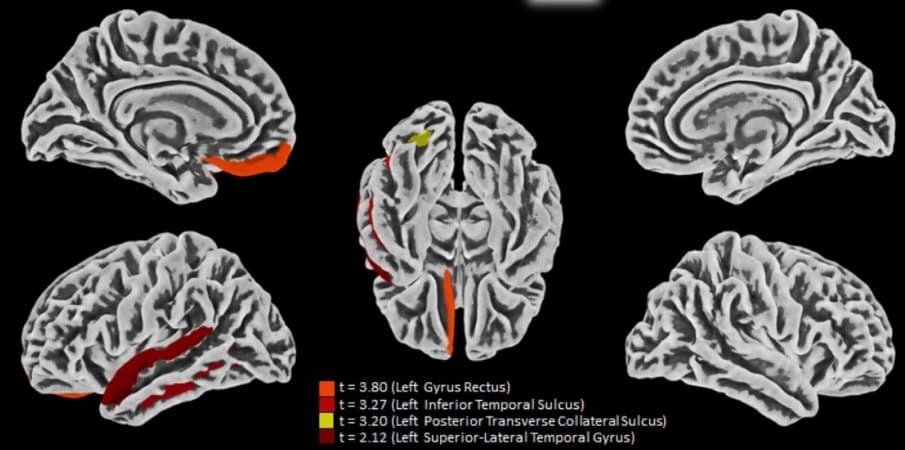
The MRI scans showed that some volunteers had decreased cortical thickness in some brain regions compared with the average for controls.
“We observed atrophy in areas associated, for example with anxiety, one of the most frequent symptoms in the study group,” Yasuda said. “Considering that the prevalence of anxiety disorders in the Brazilian population is 9%, the 28% we found is an alarmingly high number. We didn’t expect these results in patients who had had the mild form of the disease.”
In neuropsychological tests designed to evaluate cognitive functioning, the volunteers also underperformed in some tasks compared with the national average. The results were adjusted for age, sex and educational attainment, as well as the degree of fatigue reported by each participant.
“The question we’re left with is this: Are these symptoms temporary or permanent? So far, we’ve found that some subjects improve, but unfortunately many continue to experience alterations,” Yasuda said.
“What’s surprising is that many people have been reinfected by novel variants, and some report worse symptoms than they had since the first infection. In view of the novel virus, we see longitudinal follow-up as crucial to understand the evolution of the neuropsychiatric alterations over time and for this understanding to serve as a basis for the development of targeted therapies.”
Energy metabolism affected
In IB-UNICAMP’s Neuroproteomics Laboratory, which is headed by Martins-de-Souza, experiments were performed on brain tissue cells from people who died of COVID-19 and astrocytes cultured in vitro to find out how infection by SARS-CoV-2 affects nervous system cells from the biochemical standpoint.
The autopsy samples were obtained via collaboration with the group led by Paulo Saldiva, a professor at the University of São Paulo’s Medical School (FM-USP). The proteome (all proteins present in the tissue) was mapped using mass spectrometry, a technique employed to identify different substances in biological samples according to their molecular mass.
“When the results were compared with those of uninfected subjects, several proteins with altered expression were found to be abundant in astrocytes, which validated the findings obtained by immunohistochemistry,” Martins-de-Souza said.
“We observed alterations in various biochemical pathways in the astrocytes, especially pathways associated with energy metabolism.”
The next step was to repeat the proteomic analysis in cultured astrocytes infected in the laboratory. The astrocytes were obtained from induced pluripotent stem cells (iPSCs). The method consists of reprogramming adult cells (derived from skin or other easily accessible tissues) to assume a stage of pluripotency similar to that of embryo stem cells.
This first part was conducted in the IDOR laboratory of Stevens Rehen, a professor at UFRJ. Martins-de-Souza’s team then used chemical stimuli to make the iPSCs differentiate into neural stem cells and eventually into astrocytes.
“The results were similar to those of the analysis of tissue samples obtained by autopsy in that they showed energy metabolism dysfunction,” Martins-de-Souza said.
“We then performed a metabolomic analysis [focusing on the metabolites produced by the cultured astrocytes], which evidenced glucose metabolism alterations. For some reason, infected astrocytes consume more glucose than usual, and yet cellular levels of pyruvate and lactate, the main energy substrates, decreased significantly.”
Lactate is one of the products of glucose metabolism, and astrocytes export this metabolite to neurons, which use it as an energy source. The researchers’ in vitro analysis showed that lactate levels in the cell culture medium were normal but decreased inside the cells. “Astrocytes appear to strive to maintain the energy supply to neurons even if this effort weakens their own functioning,” Martins-de-Souza said.
As an outcome of this process, the functioning of the astrocytes’ mitochondria (energy-producing organelles) was indeed altered, potentially influencing cerebral levels of such neurotransmitters as glutamate, which excites neurons and is associated with memory and learning, or gamma-aminobutyric acid (GABA), which inhibits excessive firing of neurons and can promote feelings of calm and relaxation.
“In another experiment, we attempted to culture neurons in the medium where the infected astrocytes had grown previously and measured a higher-than-expected cell death rate. In other words, this culture medium ‘conditioned by infected astrocytes’ weakened neuron viability,” Martins-de-Souza said.
The findings described in the article confirm those of several previously published studies pointing to possible neurological and neuropsychiatric manifestations of COVID-19.
Results of experiments on hamsters conducted at the Institute of Biosciences (IB-USP), for example, reinforce the hypothesis that infection by SARS-CoV-2 accelerates astrocyte metabolism and increases the consumption of molecules used to generate energy, such as glucose and the amino acid glutamine.
The results obtained by the group led by Jean Pierre Peron indicate that this metabolic alteration impairs the synthesis of a neurotransmitter that plays a key role in communication among neurons.
Unanswered questions
According to Martins-de-Souza, there is no consensus in the scientific literature on how SARS-CoV-2 reaches the brain.
“Some animal experiments suggest the virus can cross the blood-brain barrier. There’s also a suspicion that it infects the olfactory nerve and from there invades the central nervous system. But these are hypotheses for now,” he said.
One of the discoveries revealed by the PNAS article is that the virus does not use the protein ACE-2 to invade central nervous system cells, as it does in the lungs.
“Astrocytes don’t have the protein in their membranes. Research by Flávio Veras [FMRP-USP] and his group shows that SARS-CoV-2 binds to the protein neuropilin in this case, illustrating its versatility in infecting different tissues,” Martins-de-Souza said.
At UNICAMP’s Neuroproteomics Laboratory, Martins-de-Souza analyzed nerve cells and others affected by COVID-19, such as adipocytes, immune system cells and gastrointestinal cells, to see how the infection altered the proteome.
“We’re now compiling the data to look for peculiarities and differences in the alterations caused by the virus in these different tissues. Thousands of proteins and hundreds of biochemical pathways can be altered, with variations in each case. This knowledge will help guide the search for specific therapies for each system impaired by COVID-19,” he said.
“We’re also comparing the proteomic differences observed in brain tissue from patients who died of COVID-19 with proteomic differences we’ve found over the years in patients with schizophrenia. The symptoms of both conditions are quite similar. Psychosis, the most classic sign of schizophrenia, also occurs in people with COVID-19.”
The aim of the study is to find out whether infection by SARS-CoV-2 can lead to degeneration of the white matter in the brain, made up mainly of glial cells (astrocytes and microglia) and axons (extensions of neurons).
“We’ve observed a significant correspondence [in the pattern of proteomic alterations] associated with the energy metabolism and glial proteins that appear important in both COVID-19 and schizophrenia. These findings may perhaps provide a shortcut to treatments for the psychiatric symptoms of COVID-19,” Martins-de-Souza pondered.
Marcelo Mori, a professor at IB-UNICAMP and a member of the Obesity and Comorbidities Research Center (OCRC), the study was only possible thanks to the collaboration of researchers with varied and complementary backgrounds and expertise.
“It demonstrates that first-class competitive science is always interdisciplinary,” he said. “It’s hard to compete internationally if you stay inside your own lab, confining yourself to the techniques with which you’re familiar and the equipment to which you have access.”
About this COVID-19 research news
Author: Press Office
Source: FAPESP
Contact: Press Office – FAPESP
Image: The image is in the public domain
Original Research: Open access.
“Morphological, cellular, and molecular basis of brain infection in COVID-19 patients” by Fernanda Crunfli et al. PNAS
Abstract
Morphological, cellular, and molecular basis of brain infection in COVID-19 patients
Although increasing evidence confirms neuropsychiatric manifestations associated mainly with severe COVID-19 infection, long-term neuropsychiatric dysfunction (recently characterized as part of “long COVID-19” syndrome) has been frequently observed after mild infection.
We show the spectrum of cerebral impact of severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) infection, ranging from long-term alterations in mildly infected individuals (orbitofrontal cortical atrophy, neurocognitive impairment, excessive fatigue and anxiety symptoms) to severe acute damage confirmed in brain tissue samples extracted from the orbitofrontal region (via endonasal transethmoidal access) from individuals who died of COVID-19. In an independent cohort of 26 individuals who died of COVID-19, we used histopathological signs of brain damage as a guide for possible SARS-CoV-2 brain infection and found that among the 5 individuals who exhibited those signs, all of them had genetic material of the virus in the brain.
Brain tissue samples from these five patients also exhibited foci of SARS-CoV-2 infection and replication, particularly in astrocytes.
Supporting the hypothesis of astrocyte infection, neural stem cell–derived human astrocytes in vitro are susceptible to SARS-CoV-2 infection through a noncanonical mechanism that involves spike–NRP1 interaction. SARS-CoV-2–infected astrocytes manifested changes in energy metabolism and in key proteins and metabolites used to fuel neurons, as well as in the biogenesis of neurotransmitters. Moreover, human astrocyte infection elicits a secretory phenotype that reduces neuronal viability.
Our data support the model in which SARS-CoV-2 reaches the brain, infects astrocytes, and consequently, leads to neuronal death or dysfunction. These deregulated processes could contribute to the structural and functional alterations seen in the brains of COVID-19 patients.