Summary: Optogentic stimulation has the potential to cultivate resilience to epileptic seizures in animal models, researchers report.
Source: Tohoku University
Around 1 percent of the world’s population lives with epilepsy; yet only 65 percent of epilepsy patients can manage their symptoms with medication.
Currently, surgically removing the lesion in the brain responsible for the condition is the only radical cure for epilepsy. Still, many patients have to take medication for the rest of their life to deal with their seizures.
A research group led by professor Ko Matsui from the Super-network Brain Physiology Lab at Tohoku University reported on a stimulation paradigm used on experimental animals that could potentially cultivate resilience to epilepsy.
Frequent seizure-evoking stimulation to the brain has been shown to induce epileptogenesis and epileptic brain conditions. To the researcher’s surprise, however, repeated stimulation resulted in a dramatic decrease in the seizure response to the stimulus.
“Our brain has an infinite ability for plasticity,” says Matsui. “If an epileptic state can be created, we must query whether it is also conceivable to reverse the transition or to override the existing hyper-excitable circuit with an additional suppressive system.”
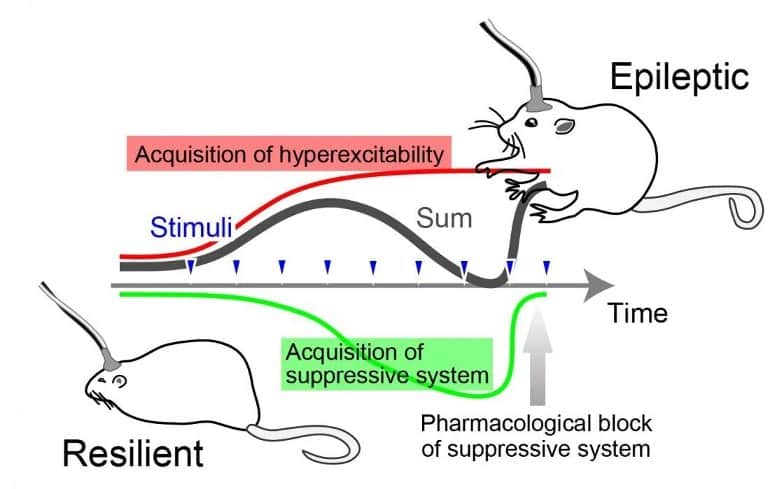
Using optogenetics technology to control the activity, Dr. Yoshiteru Shimoda, Matsui and their team demonstrated that a specific stimulation paradigm prompted the release of the endogenous inhibitory transmitter adenosine from glial cells. This converted the rat’s brain to a state strongly resistant to seizures.
Details of their findings were published in Neurobiology of Disease.
Matsui is cautiously optimistic. “Although epileptogenesis unfortunately could not be reversed, we showed we could invoke the homeostatic nature of the brain circuit to contain hyper-excitation.”
For the current study, light-sensitive proteins were genetically expressed in neurons to regulate moderate neuron-to-glial signaling at will. Such optogenetic technology would be difficult to apply in human patients, noted Matsui.
“Despite clinical use being a long way off, it is possible to imagine a future where a therapeutic strategy can directly target glial cells and enable the creation of an epileptic resistant state.”
About this epilepsy and brain plasticity research news
Author: Press Office
Source: Tohoku University
Contact: Press Office – Tohoku University
Image: The image is credited to Ko Matsui
Original Research: Open access.
“Optogenetic stimulus-triggered acquisition of seizure resistance” by Yoshiteru Shimoda et al. Neurobiology of Disease
Abstract
Optogenetic stimulus-triggered acquisition of seizure resistance
Unlike an electrical circuit, the hardware of the brain is susceptible to change. Repeated electrical brain stimulation mimics epileptogenesis. After such “kindling” process, a moderate stimulus would become sufficient in triggering a severe seizure.
Here, we report that optogenetic neuronal stimulation can also convert the rat brain to a hyperexcitable state. However, continued stimulation once again converted the brain to a state that was strongly resistant to seizure induction.
Histochemical examinations showed that moderate astrocyte activation was coincident with resilience acquisition. Administration of an adenosine A1 receptor antagonist instantly reverted the brain back to a hyperexcitable state, suggesting that hyperexcitability was suppressed by adenosine.
Furthermore, an increase in basal adenosine was confirmed using in vivo microdialysis. Daily neuron-to-astrocyte signaling likely prompted a homeostatic increase in the endogenous actions of adenosine.
Our data suggest that a certain stimulation paradigm could convert the brain circuit resilient to epilepsy without exogenous drug administration.






