Summary: Researchers report on a new method that allows linking individual neurons and brain network activity to control behavior in zebrafish.
Source: Max Planck Institute.
New method allows linking individual neurons and network activity to behaviour in zebrafish.
Scientists from the Max Planck Institute of Neurobiology in Martinsried have developed a method that allows them to identify nerve cells involved in a specific motor command. For the first time, it is now possible to evoke behaviour of a small fish by artificially activating just a handful of neurons. Understanding the core components of a neural circuit is a key step for deciphering the complex code underlying even elementary brain functions.
Recent years have seen much progress in understanding of the brain’s structure and function. Advances in microscopy and functional imaging enable researchers to monitor the activity of neuronal populations, while an animal perceives sensory stimuli or generates specific behaviours. However, these studies often cannot distinguish cause from consequence of the observed changes in activity. Using the method of optogenetics, scientists can find out which neurons are essential for the chain of events that ultimately lead to behaviour, and which neurons may serve other tasks or are merely by-standers. A particular challenge for this field of research is the staggering degree of “interconnectedness” of neuronal networks. Activating even a single neuron may send ripples through a large part of the nervous system. The new study from Herwig Baier and his team at the Max Planck Institute of Neurobiology has removed both obstacles in one sweep: it is now possible to pinpoint cause and effect to the cellular components of neural circuitry while simultaneously watching how activity propagates through the entire brain network and evokes behaviour.
The Martinsried scientists developed a workflow allowing the 3D photo-stimulation of multiple targeted neurons while, at the same time, imaging network activity in the brain of a zebrafish larva. “The zebrafish with its small, translucent brain is ideal for our new method”, explains Marco dal Maschio, one of the two lead authors of the publication describing the technique.
New approach
A few years ago, members of the Baier lab had identified a small neuronal population in the zebrafish brain whose activation triggered the bending of the tail. “This previous work did not allow a high-resolution recording of the induced activity patterns”, explains Joseph Donovan, the second lead author. “For this reason, we designed a new approach.”
First, the scientists used genetic engineering to generate zebrafish with a photo-switchable ion channel in their nerve cells. The brain activity of these animals could be remotely controlled by shining light through skin and skull into their heads. Donovan and dal Maschio then placed a zebrafish larva with such a light-switchable brain on the stage of a microscope and projected computer-generated holograms, composed of three-dimensional light patterns, into the fish brain. The light beams were in the infrared range (so-called two-photon mode), which makes them invisible to the fish, and were shaped to target a small group of individual neurons. A fast camera recorded the animal’s tail movements while the neurons were activated in changing combinations. This was repeated until a group of just three neurons was identified whose activation was sufficient to trigger the bending of the tail. The team then stimulated these three cells and simultaneously recorded the activity spreading through the brain using a fast 3D imaging approach.
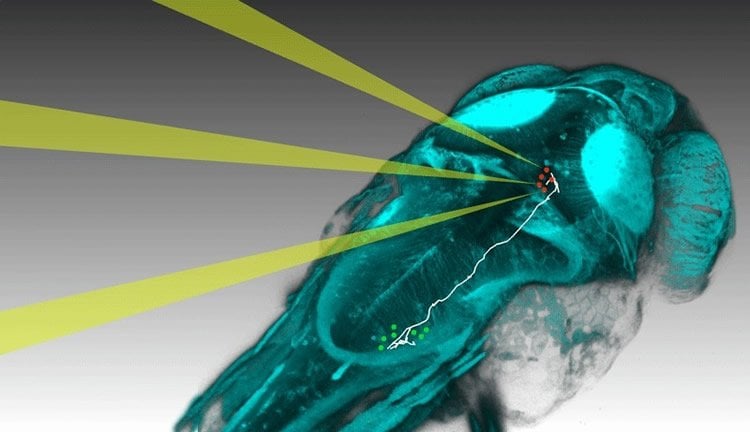
Wiring diagramm of the fish brain
From this dataset, a computer program identified activity patterns associated with distinct components of the elicited behaviour and assigned each individual neuron a “contribution score”. Finally, single neurons with interesting functions were reconstructed by visualizing their shape under the microscope. Because brains are wired very similarly from fish to fish, it is possible to draw a wiring diagram by combining data from many such experiments.
“It’s the first time that a behavioural command can be traced as it spreads from a few initial cells throughout the brain to the actual physical action”, rejoices Marco dal Maschio. He has good reason to be optimistic: The new experimental workflow lets him and his colleagues explore brain circuits in unprecedented detail. As the function and structure of the brain are conserved by evolution between fish and mammals, the new insights such studies will provide are likely to shed light on general principles of brain and behaviour.
Source: Stefanie Merker – Max Planck Institute
Image Source: NeuroscienceNews.com image is credited to MPI of Neurobiology/ dal Maschio.
Original Research: Abstract for “Linking neurons to network function and behaviour by two-photon holographic optogenetics and volumetric imaging” by Marco dal Maschio, Joseph C. Donovan, Thomas O. Helmbrecht, and Herwig Baier in Neuron. Published online May 17 2017 doi:10.1016/j.neuron.2017.04.034
[cbtabs][cbtab title=”MLA”]Max Planck Institute “Remote Control of Behavior By Optically Activating Single Neurons.” NeuroscienceNews. NeuroscienceNews, 17 May 2017.
<https://neurosciencenews.com/behavior-optogenetics-6711/>.[/cbtab][cbtab title=”APA”]Max Planck Institute (2017, May 17). Remote Control of Behavior By Optically Activating Single Neurons. NeuroscienceNew. Retrieved May 17, 2017 from https://neurosciencenews.com/behavior-optogenetics-6711/[/cbtab][cbtab title=”Chicago”]Max Planck Institute “Remote Control of Behavior By Optically Activating Single Neurons.” https://neurosciencenews.com/behavior-optogenetics-6711/ (accessed May 17, 2017).[/cbtab][/cbtabs]
Abstract
Linking neurons to network function and behaviour by two-photon holographic optogenetics and volumetric imaging
Highlights
•Simultaneous two-photon 3D imaging and 3D photostimulation
•Holographic optogenetics to explore behavior-related neuronal circuits
•Iterative approach plus analysis framework to generate/validate circuit models
•Morphological reconstruction of functionally identified neurons
Summary
We introduce a flexible method for high-resolution interrogation of circuit function, which combines simultaneous 3D two-photon stimulation of multiple targeted neurons, volumetric functional imaging, and quantitative behavioral tracking. This integrated approach was applied to dissect how an ensemble of premotor neurons in the larval zebrafish brain drives a basic motor program, the bending of the tail. We developed an iterative photostimulation strategy to identify minimal subsets of channelrhodopsin (ChR2)-expressing neurons that are sufficient to initiate tail movements. At the same time, the induced network activity was recorded by multiplane GCaMP6 imaging across the brain. From this dataset, we computationally identified activity patterns associated with distinct components of the elicited behavior and characterized the contributions of individual neurons. Using photoactivatable GFP (paGFP), we extended our protocol to visualize single functionally identified neurons and reconstruct their morphologies. Together, this toolkit enables linking behavior to circuit activity with unprecedented resolution.
“Linking neurons to network function and behaviour by two-photon holographic optogenetics and volumetric imaging” by Marco dal Maschio, Joseph C. Donovan, Thomas O. Helmbrecht, and Herwig Baier in Neuron. Published online May 17 2017 doi:10.1016/j.neuron.2017.04.034






