Summary: New research has identified the specific brain circuits that determine how we react to danger—and how those responses change as a threat fades. While fear research has traditionally focused on “freezing,” a new study reveals that the central amygdala acts as a sophisticated control center, choosing between passive freezing and active escape behaviors like “darting” or “jumping.”
By studying mice, the team discovered that fear extinction (the process of unlearning fear) isn’t about erasing a memory, but rather recalibrating which neural circuit is in charge. This “nuanced view” of fear regulation offers a biological explanation for why PTSD symptoms—ranging from hypervigilance to panic—vary so much between individuals.
Key Facts
- Beyond Freezing: The study tracked a spectrum of defensive behaviors, including freezing, escape jumping, and darting, proving that fear is a continuum rather than a simple “on/off” switch.
- The Neural Switch: Two distinct types of neurons in the central amygdala drive different reactions:
- CRF Neurons: Drive high-intensity, “panic-like” escape responses (jumping).
- SOM Neurons: Promote “lower-intensity” behaviors like freezing or darting.
- Extinction as “Reshaping”: When a threat diminishes, the brain doesn’t delete the fear; it shifts the signal from high-intensity CRF circuits to SOM-regulated states.
- PTSD Implications: Disruptions in these specific circuits may explain why some people with PTSD remain stuck in high-intensity “flight” modes while others remain in a state of hypervigilant “freeze.”
Source: Tulane University
Researchers at Tulane University have identified brain circuits that help determine how fear responses change as perceived threats diminish, offering new insight into how the brain regulates defensive behavior and why those processes may break down in conditions such as post-traumatic stress disorder.
The study, led by neuroscientist Jonathan Fadok at the Tulane Brain Institute, examines how different populations of neurons deep in the brain shape a range of fear responses – from freezing to active escape behaviors.
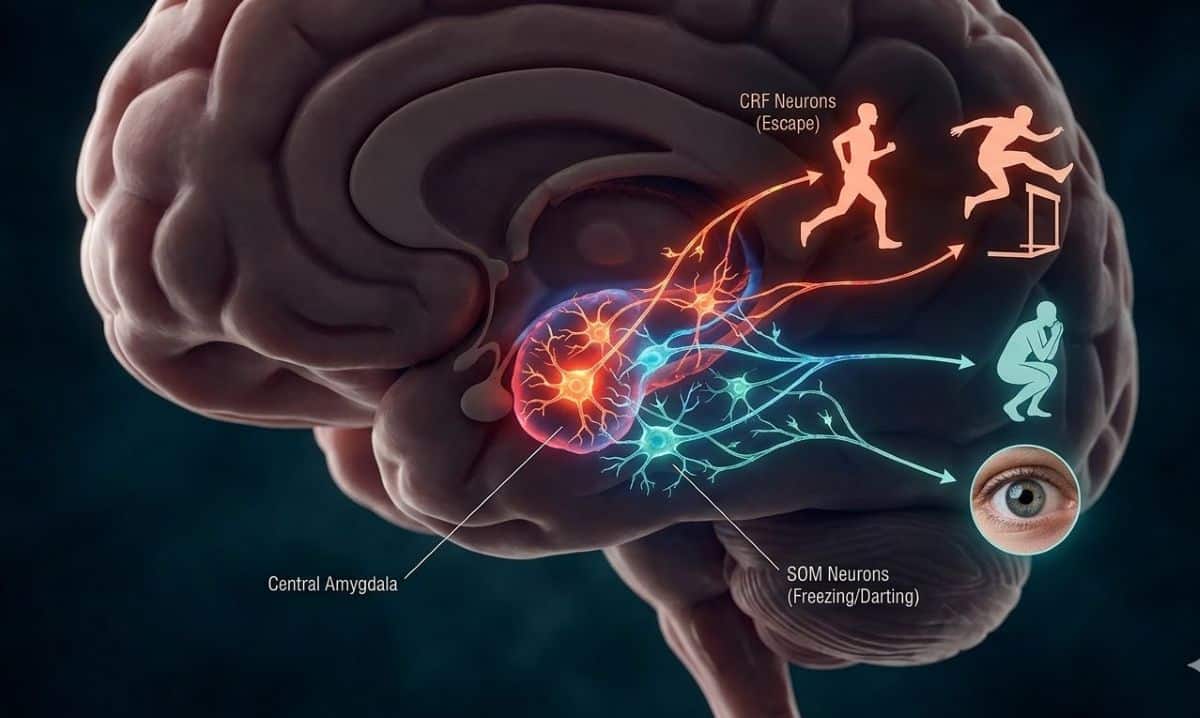
The research, supported by the National Institutes of Health and the U.S. Department of Veterans Affairs, examines how the brain adjusts fear responses as perceived threats fade.
“For decades, most fear research has focused on freezing,” said Fadok, associate professor of psychology in Tulane University’s School of Science and Engineering. “That has been incredibly useful, but it captures only part of the picture. In real-world situations, fear can also produce more active responses like darting or trying to escape.”
Using a modified conditioning paradigm in mice, the researchers were able to observe multiple defensive behaviors within the same experiment, including freezing, escape jumping and darting.
This approach allowed them to track how those behaviors shift during fear extinction – the process through which repeated exposure to a previously threatening cue reduces the fear response.
The team found that fear does not simply disappear during extinction. Instead, the brain appears to gradually recalibrate how it responds to a perceived threat.
“At the neural level, extinction looks less like erasing fear and more like reshaping it,” Fadok said. “Different circuits help determine whether an animal responds with intense escape behavior, freezing or a lower-intensity defensive state.”
Specifically, the researchers identified distinct roles for two types of neurons in the central amygdala, an area of the brain responsible for emotional processing. Activity in corticotropin-releasing factor (CRF) neurons supported higher-intensity, escape-like responses, such as jumping. In contrast, somatostatin (SOM) neurons promoted freezing and helped regulate lower-intensity behaviors like darting.
Manipulating these circuits altered how animals responded to threats. Inhibiting CRF neurons reduced escape jumping, while activating SOM neurons shifted behavior away from flight and toward freezing.
The findings suggest that the brain organizes fear along a continuum, adjusting behavior based on perceived threat level rather than switching fear “on” or “off.”
This more nuanced view of fear regulation may have implications for understanding psychiatric conditions such as PTSD, in which fear responses can be persistent and difficult to control.
“PTSD is often described as a disorder of persistent fear, but that persistence can look very different from person to person,” Fadok said.
“Some individuals remain hypervigilant, while others experience more intense, panic-like reactions. Our work points to brain mechanisms that may contribute to those different expressions.”
Although the research does not immediately translate into new treatments, it identifies biological pathways that could serve as targets for future therapies aimed at improving fear extinction.
“If extinction depends on shifting responses away from high-intensity states, then disruptions in these circuits could help explain why fear remains so hard to regulate,” Fadok said.
The study also revealed that the central amygdala plays a more active role in selecting specific defensive behaviors than previously understood.
“It’s not just generating fear,” Fadok said. “It’s helping decide what that fear looks like.”
Key Questions Answered:
A: It comes down to a “decision” made deep in your brain’s amygdala. This study shows that if your CRF neurons take the lead, you’re more likely to bolt or jump. If your SOM neurons are more active, you’re more likely to freeze in place.
A: Surprisingly, no. The research suggests your brain doesn’t actually “erase” the fear. Instead, it learns to “reshape” the response. It basically reroutes the signal from a “high-intensity panic” circuit to a “low-intensity calm” one.
A: PTSD isn’t one-size-fits-all. By identifying the exact biological “valves” that control fear intensity, scientists can work on therapies that help “nudge” the brain out of a permanent high-intensity escape state and back into a manageable, regulated one.
Editorial Notes:
- This article was edited by a Neuroscience News editor.
- Journal paper reviewed in full.
- Additional context added by our staff.
About this neuroscience research news
Author: Stacey Jenkins
Source: Tulane University
Contact: Stacey Jenkins – Tulane University
Image: The image is credited to Neuroscience News
Original Research: Closed access.
“Corticotropin-releasing factor and somatostatin neurons in the central amygdala mediate dynamic defensive behaviors during fear extinction” by Quan-Son Eric Le, Emma Lardant, Yumnah Siddiqui, Thamidul Alam, Kylie Evans and Jonathan P. Fadok. Journal of Neuroscience
DOI:10.1523/JNEUROSCI.1049-25.2026
Abstract
Corticotropin-releasing factor and somatostatin neurons in the central amygdala mediate dynamic defensive behaviors during fear extinction
Traumatic experiences can result in heightened fear responses to trauma-associated stimuli. These symptoms can be difficult to extinguish, so identifying neuronal targets for facilitating fear extinction is critical.
Many studies investigating fear learning in mice measure conditioned fear via freezing, but other defensive behaviors, such as flight, can also be present during conditioning. The central amygdala (CEA) mediates conditioned freezing and flight responses via corticotropin-releasing factor-positive (CRF+) and somatostatin-positive (SOM+) neuron populations.
However, it is unknown how these populations regulate changes in freezing and flight responses as fear extinction is learned.
Thus, we investigated the roles of CRF+ and SOM+ CEA neurons in modulating defensive behaviors during extinction. To elicit dynamic defensive responses in male and female mice, we used a modified Pavlovian conditioned flight paradigm that paired an aversive footshock with a serial compound stimulus (SCS) consisting of tone followed by white noise (WN), resulting in freezing during the tone that rapidly transitioned into flight (escape jumping and darting) during WN.
We used optogenetics in CRF-Cre and SOM-Cre mice to selectively excite and inhibit CRF+ and SOM+ CEA populations during WN presentation within extinction. Within early extinction, CRF+ inhibition reduced WN-evoked jumping and led to subsequent context-specific reduction in tone-evoked freezing.
During extinction, SOM+ excitation replaced early WN-evoked flight with freezing, and both SOM+ excitation and inhibition reduced WN-evoked darting.
Collectively, these data demonstrate modulation of jumping and darting behaviors within extinction via CRF+ and SOM+ CEA activity. These findings suggest mechanisms of attenuating multiple defensive behaviors during extinction.






