New nerve cell connections on sensory cells in mice could be at fault.
Conventional wisdom has long blamed age-related hearing loss almost entirely on the death of sensory hair cells in the inner ear, but research from neuroscientists at Johns Hopkins has provided new information about the workings of nerve cells that suggests otherwise.
In a paper published July 1 in tThe Journal of Neuroscience, the Johns Hopkins team says its studies in mice have verified an increased number of connections between certain sensory cells and nerve cells in the inner ear of aging mice. Because these connections normally tamp down hearing when an animal is exposed to loud sound, the scientists think these new connections could also be contributing to age-related hearing loss in the mice, and possibly in humans.
“The nerve cells that connect to the sensory cells of the inner ear are known to inhibit hearing, and although it’s not yet clear whether that’s their function in older mice, it’s quite likely,” says Paul Fuchs, Ph.D., the John E. Bordley Professor of Otolaryngology-Head and Neck Surgery at the Johns Hopkins University School of Medicine. “If confirmed, our findings give us new ideas for how physicians may someday treat or prevent age-related hearing loss.”
Fuchs says the new research builds on the knowledge that inside the ear lies a coiled row of sensory cells responsible for converting sound waves into electrical signals sent through nerve cells to the brain, which processes and tells animals what they “hear.” Two sets of these so-called hair cells — named for the filaments that act like antennae picking up sound waves — exist, an inner tier closest to the brain and an outer tier. The outer ones have a secondary function: to amplify the sound waves within the inner ear. Not surprisingly, Fuchs notes, a loss of outer hair cells closely correlates with a loss of hearing.
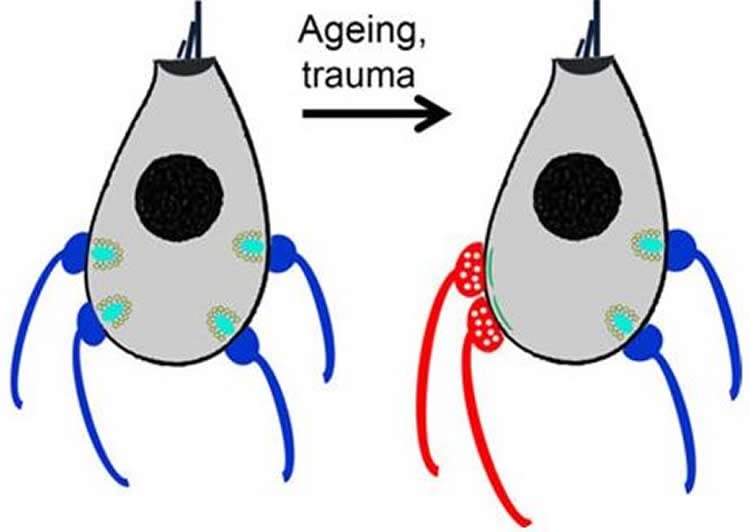
But studies over the last decade have suggested that changes over time also occur in the connections between hair cells and the nerve cells to which they are attached.
Each of those nerve cells is like a one-way street, Fuchs says, taking signals either from the ear to the brain or vice versa. The nerve cells that take signals to the ear are known to turn down the amplification provided by outer hair cells when an animal is, for example, exposed to a noisy environment for an extended period of time.
Previous research has suggested that with age, inner hair cells in mice and humans experience a decrease in outgoing nerve cell connections, while incoming nerve cell connections increase.
To find out if the new connections worked — or worked normally — Stephen Zachary, a graduate student in Fuchs’ laboratory, painstakingly recorded electrical signals from within the inner hair cells of young and old mice.
He found that the incoming nerve cells were indeed active and that their activity levels correlated with the animals’ hearing abilities: The harder of hearing an animal was, the higher the activity of its incoming nerve cells.
“These nerve cell connections seem to be reverting back to the way they worked during early development before the animals’ sense of hearing was operating,” says Fuchs. “We don’t know why the new connections form, but it might be as simple as a lack of competition for space once the outgoing nerve cells have retracted.”
If the same phenomenon is occurring in human ears, Fuchs and his team say there may be ways of preventing the incoming nerve cells from forming new connections with inner hair cells, a technique that could help maintain normal hearing through old age.
Funding: The study was funded by NIH/National Institute on Deafness and Other Communication Disorders.
Source: Catherine Kolf – Johns Hopkins Medicine
Image Credit: The image is credited to Paul Fuchs, Johns Hopkins Medicine
Original Research: Abstract for “Re-Emergent Inhibition of Cochlear Inner Hair Cells in a Mouse Model of Hearing Loss” by Stephen Paul Zachary and Paul Albert Fuchs in Journal of Neuroscience. Published online July 1 2015 doi:10.1523/JNEUROSCI.0879-15.2015
Abstract
Re-Emergent Inhibition of Cochlear Inner Hair Cells in a Mouse Model of Hearing Loss
Hearing loss among the elderly correlates with diminished social, mental, and physical health. Age-related cochlear cell death does occur, but growing anatomical evidence suggests that synaptic rearrangements on sensory hair cells also contribute to auditory functional decline. Here we present voltage-clamp recordings from inner hair cells of the C57BL/6J mouse model of age-related hearing loss, which reveal that cholinergic synaptic inputs re-emerge during aging. These efferents are functionally inhibitory, using the same ionic mechanisms as do efferent contacts present transiently before the developmental onset of hearing. The strength of efferent inhibition of inner hair cells increases with hearing threshold elevation. These data indicate that the aged cochlea regains features of the developing cochlea and that efferent inhibition of the primary receptors of the auditory system re-emerges with hearing impairment.
SIGNIFICANCE STATEMENT Synaptic changes in the auditory periphery are increasingly recognized as important factors in hearing loss. To date, anatomical work has described the loss of afferent contacts from cochlear hair cells. However, relatively little is known about the efferent innervation of the cochlea during hearing loss. We performed intracellular recordings from mouse inner hair cells across the lifespan and show that efferent innervation of inner hair cells arises in parallel with the loss of afferent contacts and elevated hearing threshold during aging. These efferent neurons inhibit inner hair cells, raising the possibility that they play a role in the progression of age-related hearing loss.
“Re-Emergent Inhibition of Cochlear Inner Hair Cells in a Mouse Model of Hearing Loss” by Stephen Paul Zachary and Paul Albert Fuchs in Journal of Neuroscience. Published online July 1 2015 doi:10.1523/JNEUROSCI.0879-15.2015






